The optimal use of genetic information to see the art and the beauty of God's Creation in Molecular Biology.
Submitted Manuscript Version
(my last additions in Red):Concise Review
Microarrays, Antiobesity and the Liver.
Fernando Castro-Chavez1
Key Words: Perilipin, Stearoyl CoA Desaturase 1, Hemoglobin, Diabetes mellitus, Inflammation.
1. Independent Biotechnologist (M.Sc.). Formerly at Baylor College of Medicine, Houston, Texas, U.S.A., at University of Guadalajara, Guadalajara, Jalisco, Mexico, and at Council of Science and Technology of Mexico (CONACyT), 72570.
E-mail: fdocc arroba yahoo.com
Abbreviations: Plin, perilipin; Scd, stearoyl CoA desaturase; Hb, hemoglobin; Srebp, sterol regulatory element-binding protein; VLDL, very low density lipoprotein; WAT, white adipose tissue.
Final Version in PDF:
Abstract:
In this review, the microarray technology and especially oligonucleotide arrays are exemplified with a practical example taken from the perilipin-/- mice and using the dChip software, available for non-lucrative purposes. It was found that the liver of perilipin-/- mice was healthy and normal, even under high-fat diet when compared with the results published for the scd1-/- mice, which under high-fat diets had a darker liver, suggestive of hepatic steatosis. Scd1 is required for the biosynthesis of monounsaturated fatty acids and plays a key role in the hepatic synthesis of triglycerides and of very-low-density lipoproteins. Both models of obesity resistance share many similar phenotypic antiobesity features, however, the perilipin-/- mice had a significant downregulation of stearoyl CoA desaturases scd1 and scd2 in its white adipose tissue, but a normal level of both genes inside the liver, even under high-fat diet. Here, different microarray methodologies are discussed, and also some of the most recent discoveries and perspectives regarding the use of microarrays, with an emphasis on obesity gene expression, and a personal remark on my findings of increased expression for hemoglobin transcripts and other hemo related genes (hemo-like), and for leukocyte like (leuko-like) genes inside the white adipose tissue of the perilipin-/- mice. In conclusion, microarrays have much to offer in comparative studies such as those in antiobesity, and also they are methodologies adequate for new astounding molecular discoveries [free full text of this article Online].
Introduction. When evaluating molecular comparative technologies such as parallel approaches1 versus serial approaches,2 it will be seen that all of them have need of improvements;3 a practical example will be provided using oligonucleotide arrays.
Recent estimates indicate that there are some 30,000 human genes and that the human proteins can reach in the million,4 while their mRNAs can be more than 134,000.5 The mRNA abundance and its mechanisms of assembly are two complementary areas. Using Intelligent Design models, we can use the analogy of the nucleic acids (DNA and RNA) as being the Software for the building up of proteins, and the proteins as being the Hardware, with the special feature that here, the Software builds up the Hardware and the Hardware plays and modulates the Software. In the case of metabolic pathways, the proteins are sufficiently flexible to become part of the regulatory Software, in a stairway of self-contained types of Hardware: organelles, cells, tissues, organs, organisms, etc.
Biological studies using solid surfaces can be done using tissue fragments disposed in rows and columns to do parallel comparisons for in situ hybridizations.6,7 Fragments of proteins and epitopes can also be aligned in the same way to be identified by labeled antibodies.8,9,10 Even cells or organelles can be arrayed.
The most used array methodology at this point is the printing of nucleic acid fragments in solid surfaces. Molecules of RNA or DNA are printed in a glass surface called the slide to produce the microarray.11 If multiple fragments of DNA are printed on nylon or on nitrocellulose, then it is called a macroarray.12 The microarrays that are made using DNA have been used mainly for gene expression comparisons under different conditions and times, but DNA microarrays can be used also to identify genetic polymorphisms between individuals.13
The original idea of doing nucleic acid massive comparisons in parallel is not new, and there are several historical antecedents for these microarray technologies, i.e., a dot-blot like procedure.1,14,15,16,17 Also, there are recent articles that describe methodological variations, like ink jet printing18, as an alternative for the commonly used pin printing, or the use of newly developed dyes with a longer life span.19
Microarray methodologies
One of the earliest microarray methodologies is on public domain and was proposed by the Stanford University,20,21,22 which uses fluorescent labeling (commonly Cy3 and Cy5 dyes) of the total RNA to be analyzed. In this methodology, many fragments of cDNA having different sizes are printed on the slide, being of 1,500 base pairs on average. Free libraries with clones for selected genes have been offered for research purposes by different scientific organizations. The main disadvantage of this methodology can be explained simply because the different size of fragments provide a variable resulting fluorescence that can be underestimating or overestimating the real presence of specific mRNAs. Additionally, the visual/manual labeling of the printed genes is highly error prone. Another disadvantage of this technology is that the fluorescent Cy3 and Cy5 dyes have a short life span, and the scanning must be done immediately after the hybridization to retrieve the best possible signal, which loses its intensity through time. Also, many of the printed fragments can correspond to the antisense mRNA strand, and the antisense probe vs. antisense targets do not hybridize. Finally, the constant update of public database annotations such as gene name, gene function, protein domain, locus, etc., require time consuming uses of internet programs like BLAST,23,24 or specialized software to verify the annotations for significant genes. Other issues involved in such cDNA microarray data analysis are given as references.25,26,27,28 Because of the reasons afore mentioned and more, after a year of unsuccessful use of this methodology, a successful switch to the technology described next was done.
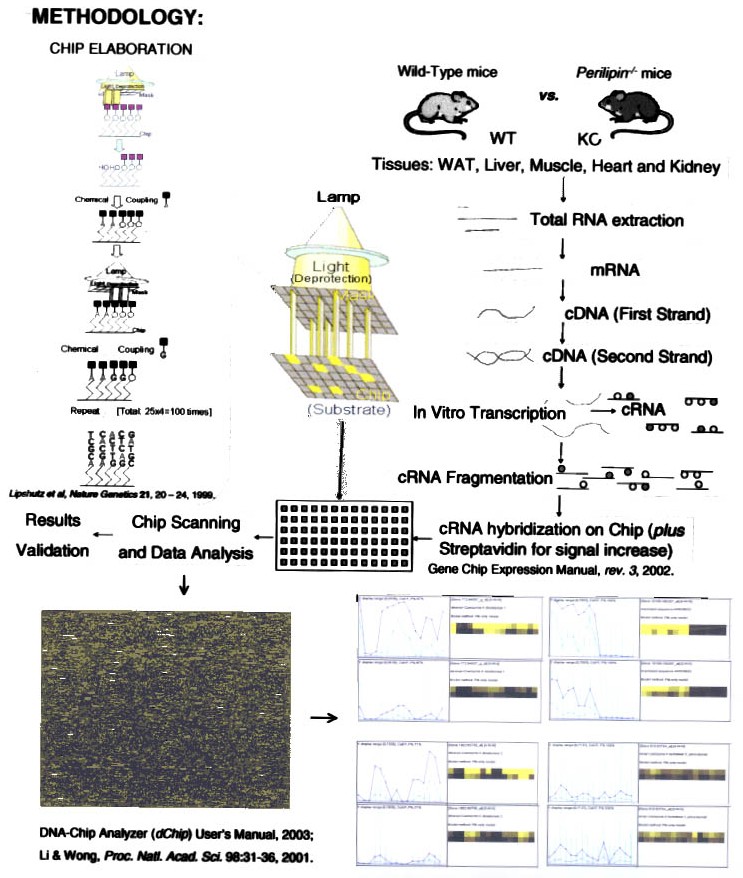
The most successful and commonly used microarray methodology is offered by the private company Affymetrix29,30,31,32,33,34 in which the fragments (oligonucleotide arrays) are synthesized on the DNA Chip, the slide. Here, DNA fragments are made by a synthesis in vitro based in Genbank sequences and using light, of which its photons are used as activators for the synthesis over specific spots uncovered by a series of masks with holes to attach one nucleotide each time. The final length of all the fragments is 25 base pairs. Each gene is represented by fragments with a perfect match (PM, commonly from 16 to 20 overlapped fragments per gene), and a replica of the same number of fragments with a mismatch (MM) at the center (the 13th nucleotide). All these fragments are printed evenly and distributed on the slide (Figure 1), then the proper software assembles them together again side by side (Figure 2). The advantage of this methodology is the uniformity of the fragments. However, if we want most accuracy, it is extremely necessary to validate the true identity of the target sequences and its probes.
If those probes include a high percent of intronic bases, then will provide a wrong estimate. One example in the mice GeneChip (MG-U74Av2) used is that there are two different Genbank IDs for the hormone sensitive lipase (hsl): U69543 and AV315398. 16 probes represent the first hsl sequence, however its last three probes are intronic (non-coding). The second hsl sequence is represented by 13 probes, with none of them being exonic (coding); therefore, the same gene name can provide two different results: a non-significant expression in the first case, or no expression at all in the second. Here it is also necessary to verify and update the names for all significant genes as both the commercial oligoarrays and the public methodology (previously described) have a high percent of sequences called ESTs (Expressed Sequence Tags), which are mRNAs for possible unknown proteins. Finally, success was attained in using this commercial oligoarray methodology and the results were published.35
A third methodology is the printing of nucleic acids on selected spots using a change in electricity from negative to positive in order to attach the DNA fragments on a solid surface.36,37,38,39 Here, the nucleic acids have a negative charge and the controlled change of polarity from negative to positive attracts them to restricted spots on the slide.
Other methodologies that have been used to compare differential gene expression are: the quantitative real time RT-PCR,40,41,42,43 subtractive hybridization,44,45,46,47 differential display,48,49,50 serial analysis of gene expression,2,5,51,52 massive parallel signature sequencing methodology,3 etc.

Figure 1. Methodology used in the microarray fabrication and application in a practical experimental design.
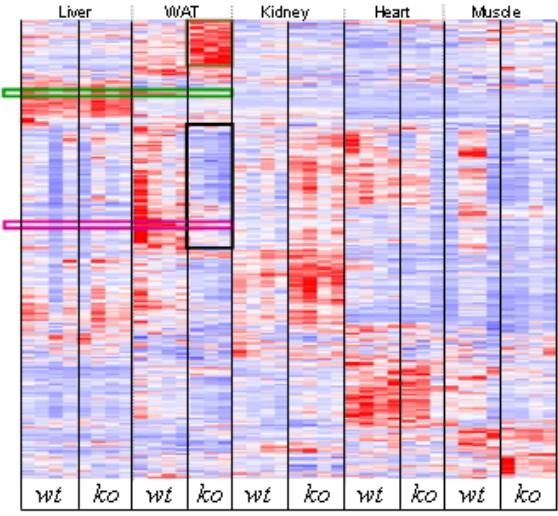
Figure 2. Hierarchical clustering of the gene expression changes between tissues of plin-/- (ko) vs. wild-type (wt) mice. Metabolic genes are upregulated in plin-/- WAT, brown rectangle. Transcription related genes are downregulated in plin-/- WAT, black rectangle, translation related genes appear below, after a short gap (not highlighted). Location of scd1, green rectangle and scd2, purple rectangle. The columns represent the organism and its tissues as labeled, and the rows represent the individual genes. A red color inside a cell in this figure represents upregulation, whereas a blue color in a cell represents downregulation. The clusters are grouped according to the intensity of expression for each particular transcript (dChip V 1.2).85 Symbols: wt, wild type (replicas); ko, knock out (replicas); WAT, white adipose tissue.
The validation of the oligoarray results was done using two commercially available quantitative real time RT-PCR methodologies (cross-validating them).35
In brief, microarrays are a useful tool to fingerprint different tissues by their specific gene expression under health and disease, and can also give us a better estimate of the ever-present genes in an organism.53,54,55
Microarrays, obesity resistance and hepatology
Examples of current discoveries with DNA microarray methodologies applied to obesity and antiobesity, with a special emphasis on the liver, will be given next. From a total of >12,400 transcripts present in mice oligoarrays, in a typical experiment, 4,867 of them are expressed by the liver.56
One of the most important discoveries done by the use of microarrays is finding that the mRNA for scd1 (stearoyl coenzyme A desaturase 1) is upregulated in the liver of obese mice, and that reaches normality after the addition of leptin, becoming a possible target against obesity.57 Surprisingly enough (as we also will see later in perilipin-/- mice microarrays), this same scd1 gene seems to be downregulated in white adipose tissue (WAT) of the ob/ob mice (Nadler et al, 2000, supplementary material),58 and its protein is absent in the naturally mutated asebia mice.57
As it is known, scd1 is an iron-containing enzyme that catalyzes a rate-limiting step in the synthesis of unsaturated fatty acids. The principal product of scd1 is oleic acid, which is formed by desaturation of stearic acid. When scd1 is downregulated, the presence of monosaturated fatty acyl-CoA, triacylglycerols, cholesterol esters and very low-density lipoprotein also goes down. Then, the fatty acids are oxidized.59,60
Another member of the same scd family, scd2 was detected with an increased expression in both the liver of the obese mice,61 and its white adipose tissue.62 The difference between obese mice microarrays and perilipin-/- mice microarrays was that in the last ones scd2 in the liver was normal, together with scd1, but in their white adipose tissue was downregulated, together with scd1.
Other tissues that had these enzymes significantly downregulated were the muscle (scd1), and the kidney (scd2).35
Human Scd1, also called Delta-9-Desaturase (ID: R00707) was found upregulated more than 2.5 fold during adipogenesis, together with PEPCK1, CD36, FABP2, Glutathione S-Transferase and Long Chain Fatty Acyl-CoA Ligase.63
Unexpectedly, with the use of oligoarrays a paradoxical finding was made: that all important genes for the fatty acid synthesis were downregulated in white adipose tissue of the obese mice, yet the plasminogen activator inhibitor 1 (pai1) was upregulated both in obese and in diabetic animals.58
Long chain fatty acyl elongase (lce), component of the elusive mammalian elongation system that converts palmitic acid into stearic acid, has been discovered in liver and adipose tissue by the use of oligoarrays in srebp transgenic mice,64 similarly the cytosolic acetyl-coenzyme A synthetase,65 and others.66
When exploring other diseases, another exciting discovery was found: In the atherosclerotic ruptured plaque, one of the most highly expressed genes was Perilipin, the same building block for the container that surrounds the big lipid droplet present inside the adipocytes.67
Mice with perilipin eliminated were able to eat fatty foods and still keep a lean body mass, but they needed to continue eating constantly in order to maintain their inner thermal and energetic balance under a mild temperature environment. If the environment is cold and perilipin-/- mice lack the proper nourishment, perilipin-/- mice will die.68
A protein found upregulated in macrophages is adipophilin (adrp), the functional precursor of perilipin in the developing adipocyte.69 Recently, the conversion of preadipocytes into macrophages has been demonstrated.70 The expression of leuko-like genes inside adipocytes of obese animals has also been reported,35,62,71,72,73 and also in obese humans.74,75 This phenomenon has been interpreted as "macrophage infiltration",76,77,78 but it is pending to discard the strong possibility of those preadipocytes being transformed into macrophages.70,79 As stated in a previous article, these findings are "pointing to the expression of common genes and regulatory pathways in these different cell types".35
In the liver of mice fed with long chain polyunsaturated fatty acids (fish oil rich in eicosapentaenoic acid and docosahexaenoic acid, plus fungal oil rich in arachidonic acid), the groups of expressed genes were linked to the transcription factors PPAR a, HNF a, and SREBP-1, all of them known to control lipid metabolism. The pattern of differentially regulated genes, supported with quantitative lipid profiling, suggested that experimental diets increased the hepatic b oxidation and gluconeogenesis while decreasing fatty acid synthesis.80 Disrupted growth hormone/insulin-like growth factor-1 signaling, which brings a phenotype of dwarfism plus caloric restriction, both extending the life-span and delaying the onset of age-related diseases in rodents, produced changes in gene expression consistent with increased insulin, glucagon and catecholamine sensitivity, gluconeogenesis, protein turnover, lipid b oxidation, apoptosis, and xenobiotic and oxidant metabolism; and on the other side, a decreased lipid and cholesterol synthesis, cell proliferation, and chaperone expression.81
In muscle and liver, adiponectin activated AMP kinase and the PPAR a pathways also increased the b oxidation of lipids, leading to decreased triglyceride content, which ameliorated insulin resistance under high-fat diet.82 The enhanced lipid b oxidation and the reduced lipid and cholesterol biosynthesis are similar features in many antiobesity mice models, like the perilipin-/- mice35 and the scd1-/- mice.83 A recent antiobesity mice model presenting similar characteristics is the mice lacking the orphan nuclear receptor estrogen-related receptor alpha.84 This finding is significant as at least 179 downregulated genes in the white adipose tissue of the perilipin-/- mice are functionally related to transcription/translation processes, representing the 33% of all the downregulated transcripts in that tissue.35 The deletion of many of those transcription/translation genes may also lead to similar antiobesity phenotypes.
An increase in the expression of the transcripts for uncoupling proteins-2 and -3 also accompanies this increase in fatty acid oxidation in the perilipin-/- adult mice, which also showed increased
b-oxidation in muscle, liver and adipose tissue. Recently has been proven that the perilipin-/- adult mice has normal plasma glucose, but a reduced basal hepatic glucose production (46% that of plin+/+).106Stearoyl-CoA Desaturase transcripts in the liver of perilipin-/- mice are normal
A variety of complementary software packages have been uploaded to the Internet for non-profit research purposes to study and interpret the massive results obtained from microarrays. The most successful and useful program freely available is called dChip,85 which provided significant results from perilipin-/- mice (plin-/-) microarray data mining.
Figure 2 shows that there were no significant changes in the liver between the plin-/- and the wild-type mice; however, when compared to the white adipose tissue (WAT), the differences were astronomical. The physical evidence showing a healthy liver inside the plin-/- mice68 also depicted only particular genes changed. These genes consisted, i.e., of the significantly downregulated steroid 21-hydroxylase and steroid sulfatase, both involved in the conversion of sulfated steroid precursors, and lysosomal acid lipase 1 and cytochrome P450 2b10,86 whereas two different transcripts for cytochrome b-245 a polypeptide were significantly upregulated inside the liver of the plin-/- mice. In total, 72 genes were downregulated and 137 genes were upregulated in the plin-/- mice liver, 30 of those upregulated genes were involved in signaling processes.35
Hierarchical clustering of five different tissues (WAT, liver, heart, muscle and kidney), when put together side by side (Figure 2), from both plin-/- and wild-type mice, revealed the following genes as coordinately regulated: long-chain fatty acyl elongase and Scd2, whereas pyruvate dehydrogenase kinase 1, aldehyde dehydrogenase 1A4 and eukaryotic translation initiation factor 1A clustered together to Scd1.35
In contrast to the normal appearance and behavior of the plin-/- mice liver under high fat diets, the liver of the scd1-/- mice on a high-fat diet had a darker color, suggesting hepatic steatosis, while its wild-type control, under the same diet, had liver with a lighter color.83 Other phenotypical characteristics for obesity resistance between these antiobesity mice models are highly similar.35
In human nonalcoholic steatohepatitis, significantly downregulated were genes important for maintaining mitochondrial function, like copper/zinc superoxide dismutase, aldehyde oxidase, catalase, glucose 6-phospatase, alcohol dehydrogenase, elongation factor-TU, methylglutaryl CoA, acyl CoA synthetase, and oxoacyl CoA thiolase, whereas genes overexpressed included complement component C3 and hepatocyte-derived fibrinogen-related protein, potentially contributing to an impaired insulin sensitivity.87 A two-hit model has been proposed in the pathogenesis of non-alcoholic steatohepatitis: First, hepatic steatosis, where a hypercaloric diet with high levels of carbohydrates and of saturated fatty acids results in elevated plasma free fatty acids and expands the adipose tissue, developing insulin resistance and augmenting even more the steatosis, plus oxidation of free fatty acids yielding toxic free radicals with the resulting lipid peroxidation; Second, increased oxidative stress on hepatocytes, and induction of pro-inflammatory cytokines. When the antioxidant capacities of the liver are insufficient, appears mitochondrial dysfunction, and TNF a cause inflammation, fibrosis and cirrhosis.88,89,90
As we have seen before, fatty acid oxidation is enhanced in the absence of scd1, required for the biosynthesis of monounsaturated fatty acids and which plays a key role in the hepatic synthesis of triglycerides and very-low-density lipoproteins.59 Triglyceride synthesis was dramatically reduced in the liver of scd1-/- mouse fed a lipogenic diet when compared to the normal mouse, but the level of cholesteryl esters of saturated fatty acids was increased.60 In an impaired triglyceride synthesis, oleate induces lipotoxicity but in the opposite side, unsaturated fatty acids serve a protective role against lipotoxicity, promoting triglyceride accumulation and preventing the palmitate-induced apoptosis.91
The previously mentioned enzyme AMP-activated protein kinase, the metabolic sensor that regulates lipid metabolism during increased energy expenditure, is 40% upregulated in liver of scd1-/-, concomitant with decreased intracellular levels of malonyl-CoA, which lead to a 60% increase in the activities of the enzymes cpt1 and cpt2, thereby stimulating the oxidation of the mitochondrial palmitoyl-CoA.92 Recently, also a plasminogen-like protein has been found that degrades scd1 in liver.93
Perspectives and conclusion
With microarrays also was found that the gene spot14 was downregulated -4.6 fold in obese mice.58 Spot14 pertains to a cluster downregulated by leptin through srebp, both in liver and in white adipose tissue,57,62 spot14 was also the gene most upregulated (8.8 Fold) by thyroid hormone in liver,94 and was increased, together with ATP citrate-lyase and fatty acid synthase in the insulin receptor substrate-2-/- mice liver.95 The significance of these differences may be understood in the future for genes with defined patterns of expression such as spot14.
New pathways can be discovered with the use of microarrays, and new functions found for known proteins. For example, hemoglobin was found to be upregulated in three different organs under three different conditions: 1-) in spleen cells of non insulin-dependent mice NOD-like, hba1 was upregulated with other 34 erythrocyte-specific transcripts;96 2-) in liver of the transgenic Scavenger Receptor-1 mice, beta globin was upregulated 1.7 fold and alpha globin 1.8 fold, the authors think that it is was due to a capture of erythrocytes mediated by an unknown liver receptor;97 3-) in the white adipose tissue of the perilipin-/- mice hbb-b1 was significantly increased (1.74 fold),35,73 and in diabetic and obese mice, hba1 was increased 4 and 5 fold, respectively (supplementary material, Nadler et al, 2000).58
Northern Blots were performed of hba1 transcript expression, finding it highly expressed in white adipose tissue, heart and kidney. Hba1 was also expressed in liver and in muscle of normal mice.73
In Gabrielsson et al (2000)55, two of his 136 most abundant mRNAs expressed in white adipose tissue of healthy humans, analyzed through DNA arrays, were hemoglobin beta and hemoglobin beta H. Planat-Benard et al (2004)98 confirmed that adipose tissue contains progenitor cells with angiogenic potential, as dedifferentiated mature human adipocytes had the potential to rapidly acquire the endothelial phenotype in vitro and to promote neovascularization in ischemic tissue with vessel-like structure formations. Those dedifferentiated adipocytes spontaneously expressed the endothelial cell markers cd31 and von Willebrand factor when cultured in a semisolid medium. Recently Kolonin et al (2004)99,100 reported a possible antiobesity treatment based on the induction of apoptosis in the vascular system of white adipose tissue through its resident peptide CKGGRAKDC, a peptide associated to prohibitin, which is a mitochondrial membrane multifunctional protein (Ikonen et al, 1995).101 Targeting the proapoptotic peptide to prohibitin in the adipose vasculature caused ablation of white fat without any detectable adverse effects in mice.
These perspectives show that, on using microarrays, new genes (i.e., spot 14) can be found, but also well known genes being expressed in unexpected tissues or cells; i.e., my findings of hbb-b1 and hba1 expressed inside white adipose tissue, and the heart,73 fulfilling their own and particular function inside a specific environment; i.e., inside a cellular organelle. Species-specific genes can also be found (i.e. mouse AA185432, unpublished results).
Because of the additional post-transcriptional editing and regulation, we can conclude that the data that we are obtaining with microarrays of nucleic acids (DNA and RNA), as a map, can explain only one important aspect of molecular biology, and should complement other methodologies able to characterize its resulting proteins, like kinetics and its maturation quality control.102,103,104 Stringent quality control needs to be enforced also in Genbank sequences, to be more confident of the results offered by microarray technologies, i.e., as oligoarrays (Affymetrix) base their targets on them. I found with microarrays an artificial phenomenon of heterotranscription contaminating thousands of sequences in the Genbank nucleic acids database through the sequence CTCGTGCCGAATTCGGCACGAG or its derivatives.105
The results of performing a careful microarray analysis of the genetic program of organisms have been helpful, thus far, in setting an integrated foundation for nutrigenomics (effects of nutrients on the genome and on the regulation of gene expression),107 and nutrigenetics (effect of genetic variation on the interaction between diet and disease and on the healthy response to nutrients)108 in developing stringent cross-validation systems (i.e., through the combined use of microarrays, and of Q-RT-PCRs,109) and to better describe the identity and function of genes110 and genomes,111,112,113 composers of a natural, complex, and precise biological software that as a genetic program, contributes to the healthy programming114 and the pathological reprogramming of life.115
Fernando Castro-Chavez wishes to thank God, and to his wife Tracy Lynn Duncan, for help in preparing this manuscript. Vijay K. Yechoor suggested the elaboration of the figures that were originally presented in a poster for an Internal Department of Medicine’s Research Symposium on Apr. 3, 2003, at Baylor College of Medicine, Houston, Texas.
References:
Other Related Links:
Research on Intelligent Design
Tasters of the Word (YouTube), videos recientes: "Astronomía y Nacimiento de Jesucristo: Once de Septiembre Año Tres A.C.", "Estudio sobre Sanidades" (en 20 episodios), "Jesus Christ, Son or God?" and "We've the Power to Heal":
Tasters of the Word (the blog, with: "Astronomy and the Birth of Jesus Christ"):