PHASE II Clinical Trial
Efficacy
and Safety of 50% Akapulko Lotion Compared to Placebo and 25% Sodium Thiosulfate
among Patients with Tinea Versicolor;
A Double Blind Randomized Controlled Clinical Trial
ABSTRACT
One hundred seventy four diagnosed cases of Tinea versicolor were entered into a double blind randomized controlled clinical trial comparing the efficacy and safety of 50% Akapulko lotion, 25% sodium thiosulfate and placebo. A total of 30 patients per treatment regimen completed the 4 week observation period. There were 51 females and 39 males and the mean age was 31.4 years old. Patients were observed weekly and the following parameters were noted: (1) mycologic cure; (2) erythema; (3) scaling; and (4) adverse reactions.
Results showed that there was a significant difference between sodium thiosulfate and Akapulko versus placebo in attaining a negative KOH mount and causing the disappearance of erythema and scaling. There was no significant difference between the three drugs in producing adverse drug reactions.
INTRODUCTION
Akapulko,
scientifically known as Cassia alata (L) or Herpetica alata (Ref), is a course,
erect, branched shrub growing up to 3 meters in height. It is locally abundant
throughout the
Traditionally, quack doctors use the leaf juice for superficial fungal infections like Tinea versicolor, Tinea circinata and Tinea pedis. In a study done by Nelia Maramba, M.D. comparing sulfur ointment and Akapulko, results showed that both Akapulko in 3 concentrations and sulfur ointment produced partial cure after 3 - 4 weeks.
Experiments have isolated chrysophanic acid as one of the active components of Akapulko lotion. It is an active reducing agent in its pure form.
Tinea versicolor is a superficial, chronic and recurrent fungal infection of the stratum corneum caused by the filamentous form of Pityrosporum orbiculare. It is clinically characterized as hypo-pigmented or hyper pigmented macules usually located on the trunk, neck and upper extremities. Present management consists of topical anti-fungal agents namely, imidazole , 25% sodium thiosulfate, sulfur-salicylic acid preparations and 50% propylene glycol. Systemic ketoconazole or itraconazole is given for widespread or resistant cases.
All of the above mentioned drugs are expensive and not readily available to patients, specially in the rural areas. Hence, this study will provide a cheaper, effective and endogenous substitute for the expensive anti-fungal preparations.
OBJECTIVES OF THE STUDY
To determine the percent cure of Akapulko as compared to 25% sodium thiosulfate
and placebo given on a twice daily application for 4 weeks.
To determine the incidence of adverse reactions on patients given Akapulko lotion, placebo and 25% sodium thiosulfate.
STUDY DESIGN
Double blind randomized controlled clinical trial.
MATERIALS AND METHODS
A. Patients Population
Inclusion Criteria:
1.
Patients consulting at the PGH Section of Dermatology Clinic for skin lesions
diagnosed clinically and by KOH mounts as Tinea versicolor.
2. Patients with minimum aggregate size of 4 cm2
3. Patients with maximum aggregate size of 20 cm2
4. Patients with no topical or systemic anti-fungal treatment prior to treatment
with assigned drugs
5. Age 15 - 60 years old
6. Written and signed informed consent
Exclusion Criteria:
1.
Pregnant and lactating mothers.
2. Any infection / super-infection of the skin requiring additional treatment
other than the trial drug except systemic anti-histamines.
3. Any systemic disease or condition predisposing a patient in developing
fungal infection
4. Poor compliance to previous topical treatment.
5. Patients with known hypersensitivity to parabens and chrysophanic
acid.
PROCEDURE
Qualified patients based on the inclusion criteria were randomly given either Akapulko lotion, 25% sodium thiosulfate or placebo. Double blind procedure was observed. Patients are to apply the assigned treatment to all affected areas using a piece of gauze twice daily and were required to follow-up on a weekly basis for a total of 4 weeks.
Patients were given a supply of bath soap to be used in the duration of the study to prevent bias with use of medicated soaps. A site for evaluation accessible to the patient to ensure drug compliance was assigned from any of the following: (1) anterior trunk; (2) face; (3) anterior neck; and (4) anterior aspect of the thigh. The size of the area for evaluation was limited to 4 x 4 cm2 and the patients were evaluated on the same site weekly. Drugs were dispensed also on a weekly basis. Discontinuance of the drug was done if any of the following were noted: (1) any adverse reaction in the form of unbearable pruritus, burning, inflammation and irritation; (2) development of intercurrent illness which will interfere with the study design or evaluation of the study; (3) failure of the patient to follow instructions given on the first visit; (4) patients request to withdraw from the study.
The
following parameters were assessed on the patients' weekly follow-
up:
(1) mycologic cure assessed as either positive or negative KOH mount;
(2) grading of the clinical signs of activity of the disease and the
keratolytic effect of the drugs in the form
of erythema and scaling as follows:

(3) presence or absence of adverse reactions such as pruritus,
burning, inflammation or irritation was monitored
every clinic visit.
STATISTICAL ANALYSIS OF DATA
Null
Hypothesis:
(1) There is no significant difference in the percent mycologic cure and disappearance
of erythema and scaling with the use of Akapulko lotion,
placebo and sodium thiosulfate in the management of Tinea versicolor
(2) There is no significant difference in the incidence of adverse reactions
in patients treated with Akapulko lotion, placebo
and sodium thiosulfate.
For Hypothesis 1 and 2, Chi-square test was used.
RESULTS
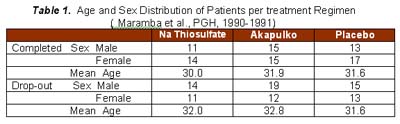
A total of 174 patients were included in the study, however, only 90 completed the 4 week observation period. All the patients were randomly assigned to different treatment regimen. Table 1 shows the distribution of the patients to the different treatment. Of the 90 patients who completed the study, there were 51 females and 39 males, and the mean age was 31.4 years .
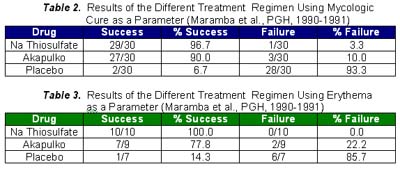
Of the 30 patients who received sodium thiosulfate, 29 (96.7%) became KOH negative in an average of 2.3 weeks after the initiation of treatment. Only 1 patient persisted to have positive KOH mount at the end of the 4 week observation period. This was comparable to those who received 50% Akapulko lotion wherein 27 (90%) had KOH negative mounts in an average of 3 weeks. For the patients who received placebo, however, only 2 (6.7%) patients had KOH negative mounts at the end of 4 weeks. This was obtained in an average of 2.5 weeks. Chi-square analysis showed a significant difference between the three treatment regimens with a p value < 0.05.
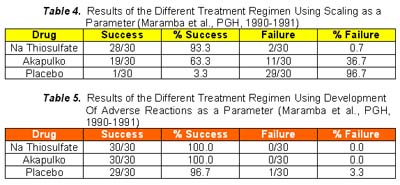
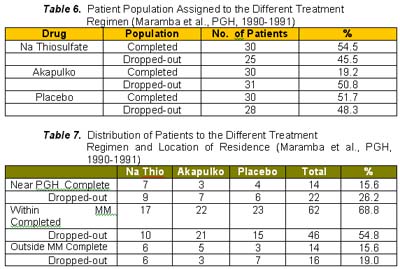
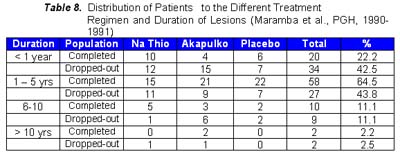
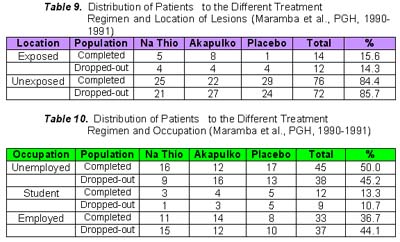
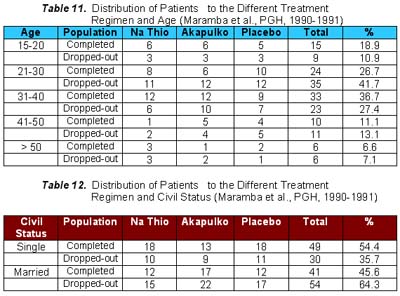
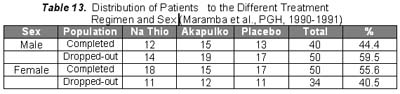
Ten of the 30 patients who received sodium thiosulfate presented with erythema and all noted the disappearance in an average duration of 2.6 weeks. Again, this was comparable to the results obtained in patients who received Akapulko lotion since 7 of 9 patients (77.8%) had disappearance of the erythema in an average of 3.6 weeks. Again, the above-mentioned results were different from those obtained in the placebo group. 7 of the 30 patients presented with erythema and only one (14.3%) noted disappearance after 1 week. Chi-square analysis showed a significant difference between the three treatment regimen with a p value < 0.05. All the patients who received sodium thiosulfate presented with scaling and 28 (93.3%) noted its disappearance in an average of 3.1 weeks. All the patients who received Akapulko also presented with scaling but only 63.3% noted its disappearance in 3.2 weeks. All patients who received placebo also presented with scaling but only 1 (3.3%) noted its disappearance after 1 week. Chi-square analysis showed a significant difference between the three treatment regimens with a p value of < 0.05. None of the patients who received sodium thiosulfate and Akapulko lotion developed adverse drug reactions. One patient in the placebo group, however, developed mild pruritus over the area of application, but was immediately relieved by anti-histamines. Statistical analysis using Chi-square analysis revealed no significant difference between the three treatment regimens with a p value > 0.05. There was a total of 84 drop-outs in this study. As shown in Table 6, 45.5% of those who received sodium thiosulfate, 50.8% of those who received Akapulko lotion and 48.3% of those who received placebo dropped-out. Possible reasons were surmised regarding the high drop-out rate, thus, patients who failed to follow-up were categorized into the different parameters that may have influenced them to drop out, namely: (1) drug received; (2) location of residence; (3) duration of lesions; (4) location of lesions; (5) occupation; (6) age; (7) civil status; and (8) sex. Those who completed the study were also categorized according to the same parameters and the results were compared. Results did not show any difference between the subject population who completed the study and those who dropped-out. Reasons for a high drop-out rate are, therefore, neither of the above mentioned reasons.
DISCUSSION
Tinea versicolor is a common dermatologic disease entity that tends to be
chronic and recurrent. Diagnosis is facilitated with the use of KOH mount
and treatment response may easily be evaluated by this method. In this study,
we designate this to be the mycologic cure. This study shows that there is
a significant difference between sodium thiosulfate and Akapulko versus placebo.
There is only a slight difference in the success rate with use of sodium thiosulfate
(96.7%) compared to Akapulko (90.0%) in the attainment of mycologic cure and
in a slightly shorter span of time. Only one patient, however, among those
who received placebo attained mycologic cure.
Erythema and scaling may or may not be present in patients with Tinea versicolor. If present however, it may be a clinical gauge of the efficacy of the treatment. Scaling is particularly important because of the capacity of the fungus to invade and inhabit the stratum corneum. Keratolytic agents, even in the absence of fungistatic or fungicidal capability alone, may prove to be effective agents in the promotion of cure of this disease. In this study, there is again a significant difference between sodium thiosulfate and Akapulko versus placebo. Sodium thiosulfate is more successful than Akapulko in causing the disappearance of scaling and erythema primarily because of its inherent characteristic as a keratolytic agent, thus the success rate of 93.3%. Akapulko also has keratolytic properties as evidenced by the 63.3% success rate but this is not as good as that seen in sodium thiosulfate. Placebo again had a very low success rate in the promotion of disappearance of both scaling and erythema.
All three drugs did not produce significant adverse reactions. In the whole study, there was only one patient who complained of mild pruritus over the areas were placebo was applied. This, however, was easily controlled by intake of anti-histamines.
CONCLUSIONS
Akapulko is an effective drug in reversing KOH findings in Tinea versicolor. It is as effective as sodium thiosulfate and is superior to placebo.
RECOMMENDATIONS
· Inclusion
of a bigger subject population to produce significant conclusions
regarding superiority of one drug over the others.
· Regularity in the drug supply to prevent disruption of randomization of
the study.
· Patient education is a must to ensure good compliance and follow-up of the
subjects.

 |
|
|

|
|
Copyright © 2002 Altermed Corporation. All rights reserved.