7.4 PHASE III CLINICAL TRIAL OF Blumea Balsamifera, L (SAMBONG) TABLET IN THE TREATMENT OF URINARY TRACT STONES: A RANDOMIZED DOUBLE-BLIND PLACEBO CONTROLLED STUDY.
7.4.1 Abstract
Sambong (Blumea balsamifera,L) tablets as Anti-Urolithiasis
The general objective of the clinical trial was to compare the efficacy and safety of sambong and placebo tablets, given at 40 mg/kg BW in three divided doses daily among 22-60 year old patients with urinary tract stones (> 5 mm at its greatest axial diameter) documented in excretory urograms. Forty subjects were screened and 19 qualified with 13 subjects completing the study, with 6 dropouts. There were 13 males and 6 females; ratio of 2.1:1. Majority of the patients were between 31 and 50 years of age. Duration of illness ranged from within 30 days of enrollment into the study (N=9), 2 within several months and in 3 patients, several years duration. The chief complaints were moderate to severe pain and signs/ symptoms referable to urinary disturbance. The most commonly used drugs were antimicrobials, analgesics or anticolic agents. Excretory urogram demonstrated solitary stone in ten patients while the rest had multiple stones. There were eleven subjects in the sambong group who completed the study and two in the placebo group. All the dropouts were in the placebo group.
7.4.2 Results
In the sambong group, 1 patient showed normal post-treatment KUB-IVP, 2 passed out stones and 2 other patients excreted sandy urine while another patient demonstrated decrease in size and number of stones. A total of 6 patients (54%) demonstrated radiographic or factual excretion of stones/ sandy materials in the urine. No patient in the placebo group demonstrated these objective improvements. Clinical response to treatment based on improvement of pain and urinary disturbance was seen in 8 patients who completed the placebo treatment continued to complain of flank pains and urinary difficulties.
7.4.3 Conclusion
a. Urolithiasis occurs more commonly among males with a 2.1 : 1 male to female ratio between the 3rd and 5th decades of life with variable duration of illness.
b. Urinary tract stones usually manifest as moderate to severe pain and disturbances of urination.
c. Solitary stone was common (2:1) compared with multiple stones based on excretory urograms of patients.
d. Sambong treatment resulted in disappearance of stone in one patient, decrease in the size and number of stones in another, actual passage of stones in 2 and excretion of sandy materials in another 2 patients with clinical improvement in all 6 patients (54%) while 2 other patients showed relief of pain and urinary disturbance for an over-all efficacy of 72 % compared to nil for placebo.
e. Dropout rate was very high among the placebo treated patients.
7.4.4 Recommendation
A phase III open-clinical trial of sambong should be conducted because of ethical problems with the use of placebo wherein the subjects with moderate to severe pain had to be given “ rescue analgesics” to provide relief for their sufferings.
7.4.5 Introduction
Blumea
balsamifera,L (Sambong) tablets have been clinically validated as a diuretic
with potassium-sparing effects and effective in Class I and II congestive
heart failure patients. The phase II study comparing placebo and sambong tablets
in 23 patients with urolithiasis given 40 mg/ day for 4 weeks characterized
the Filipino subjects as clustering between the 3rd to 5th decades of life,
with a 1.5: 1 male to female ratio, and the most common presenting complaint
as abdominal pain and those referable to urinary problems with 48% reporting
urinary tract infection, treated with antibiotics. Hyperurecemia ranked first
among the associated abnormalities. Majority of the patients had solitary
stone (80%) more than 5 mm in size mostly radio-opaque. Results of treatment
showed that both placebo and sambong therapy did not alter significantly urinary
volume, pH, protein, creatinine, as well as urinary and serum uric acid and
calcium. Radiographic evidences of decrease in size of stones and the combined
decrease in the number and/ or size of stones occurred in 77% of subjects
compared with 10% in the placebo group which was statistically significant.
There was no difference in the reported adverse effects between placebo and
sambong as to frequency and severity. The recent study of Rico et al showed
urinary chemolysis of calcium stones when sambong was given at 40 mg/kg/day.
Although the management of urinary calculi has been greatly revolutionalized
by the use of Extracorporeal Shock Wave Lithotripsy (ESWL), many stone formers
could not yet avail of this technology due to prohibitive cost and availability
only in tertiary hospitals.
The promising results in the Phase II clinical trial of sambong tablet for urolithiasis may provide the therapeutic modality that is affordable, non-invasive and accessible to most Filipinos.
7.4.6 Objective
General Objectives:
1.
To compare the efficacy and safety of Blumea balsamifera, L. tablet (Sambong)
against placebo on spontaneous stone passage, and/or decrease
in number and/or size of stones.
2. To compare the effect of sambong tablet vs. placebo on renal colic and
other urinary tract symptoms secondary to urolithiasis.
3. To determine the adverse effects of sambong tablets when administered to patients with urinary calculi.
4. To determine the acceptability of sambong tablet vs. placebo in patients with urolithiasis.
METHODOLOGY
Subjects
Patients will be selected from among those consulting at the Out-Patient facilities of the: Philippine General Hospital,
Patient inclusion criteria:
1. male of female patients with age ranging from 22-60 years.
2. Patients with urinary tract stones >5 mm (greatest axial diameter)
3. With normal renal function
4.
Willingness to participate in this study by signing the study consent
form
Patient Exclusion Criteria:
1. With nephrocalcinosis or bladder outlet obstruction
2. With severe urinary tact stone-associated symptoms
3. With acute pyelonephritis, glomerulonephritis or renal parenchymal diseases
4. With any of the following diseases: hyperthyroidism, other hypercalcemic disorders, distal renal tubular acidosis, uncontrolled NIDDM, insulin-dependent DM or acute gouty arthritis.
5. With moderate to severe hypertension, bronchial asthma, chronic liver disease, history of severe allergy, chronic or acute diarrhea.
6. With intake within 2 weeks of consultation of diuretics, allopurinol acetazolamide, ascorbic acid.
7. With intake of short acting or intermediate acting corticosteroids within one month and six months of consultation respectively.
7.4.7 Procedure
Baseline and follow-up evaluations of patients include thorough clinical history, physical and laboratory examinations with emphasis on urinary tract assessment.
Instructions
on diet, fluid intake and intake of medications will be emphasized to patients.
Patients shall be randomly assigned to one of the two treatment groups: Sambong
tablets (250mg) at a dose of 40 mg/kg/day and equivalent dosing using placebo
(250 mg/tablet)
Sample size will be computer as follows:
If alpha = 0.05 Beta = 0.1
Success rate
Sambong 0.7
Placebo 0.25
N = 24/group
(evaluated). If 25% allowance for drop-out is computed, N = 30/group
Since only 50% those screened, qualify for admission, at least 60/group or
total of 120 subjects will be screened.
Details of follow-up visit monitoring, are incorporated in the patient case forms. Patients who do not pass out stones or did not improve clinically or radiographically considered evaluable if baseline data is complete and has either passed out stones, or completed the 6-week treatment with post-treatment evaluation.
7.4.8 Results and Discussion
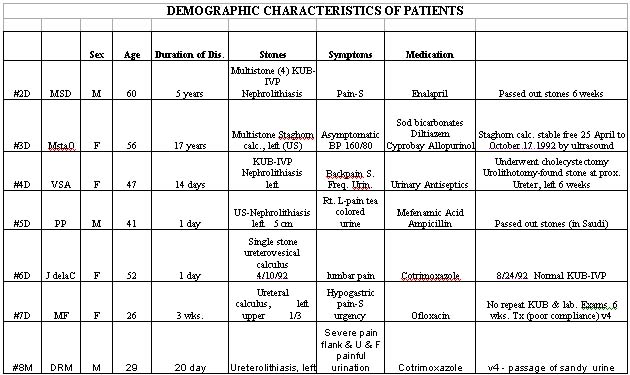
A total of 19 subjects were enrolled into the study after 40 subjects were screened. Thirteen subjects have completed the treatment and 4 dropped-out. There are at present 2 subjects on follow-up. There were 13 males and 5 females among those enrolled with male to female ratio of 2.1:1. Majority of the subjects enrolled were between 31- 50 years of age. There were 9 subjects whose disease duration was within 30 days of consultation, 2 within several months and 3 within the duration of several years. The main complaints were moderate to severe back pain, frequency and urgency of urination, dysuria and gross hematuria. Previous medications taken were mostly antimicrobials, analgesics or anti-colic. Ten subjects showed single stone while 4 showed multiple stones. There were 11 subjects in group 2 and only 2 subjects in group 1 who completed the study. There were 6 patients who responded by either passing out stones (N=1) or by passing sandy urine (N=2). One patient who cholecystectomy and also urolithotomy showed that the urethral stone had passed from the calyces to the ureter. There were no spontaneous passage or radiologic evidence or decrease in stone size or number in group 1. Subjects in the two groups found the tablets acceptable.
7.4.9 Conclusion
There is a difference in response between the two groups. Six out of 11 patients in the sambong group responded by passing out stone (N=2), passed sandy urine with relief of lumbar pains (N=1), normal repeat KUB with disappearance of stone (N=1); descent of stone from calyces to ureter (N=1) and decrease in number of stones.

SAMBONG AS ANTI-UROLITHIASIS CLINICAL TRIAL
7.5 EXTENDED PHASE III OPEN TRIAL OF Blumea balsamifera, L (SAMBONG) – FOR THE TREATMENT OF URINARY TRACT STONES
7.5.1 Abstract
The open-clinical study was undertaken to evaluate the efficacy and safety of orally taken sambong tablets on spontaneous stone passage, decrease in size and number of urinary stones and clinically evaluable signs/ symptoms of urinary disturbance. Patient inclusion criteria were: presence of urinary stone >5mm in greatest axial diameter documented by excretory urogram or renal ultrasound among 22-60 year old patients and were otherwise with normal organ function who signed the informed consent.
A total of 40 patients were screened and 18 were found qualified to enter the clinical trial. Sambong tablets were given at 40 mg/kg BW in three divided doses daily together with total intake of at least 3 liters of water daily for a total of 6 weeks of therapy.
Sociodemographic and clinical profile of patients were as follows:
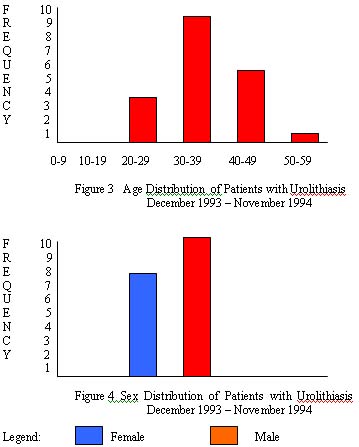
1. patients with urolithiasis clustered at the 3rd to 4th decade of life, with a mean age of 37 years;
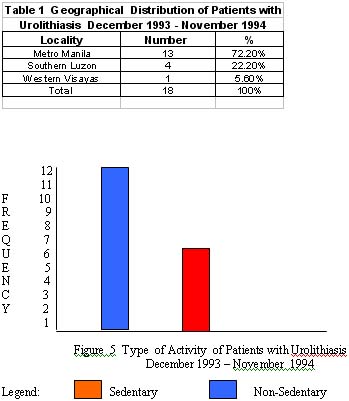
2. a male to female ratio of 1.56:1 was seen with sedentary life style in majority of patients and preference for salty foods (fish and soy sauce condiments) and low fluid intake (1-1.5 liters/day);
3. the most common presenting complaint is flank pain / CVA tenderness (61.1%) followed by signs and symptoms of urinary disturbance ( hematuria, urgency and dysuria);
4. duration of illness varied from several months (77%) to several years (22%);
5. Antimicrobials, uricosuric drugs and antispasmodics were the most commonly used medications prior to entry into the study;
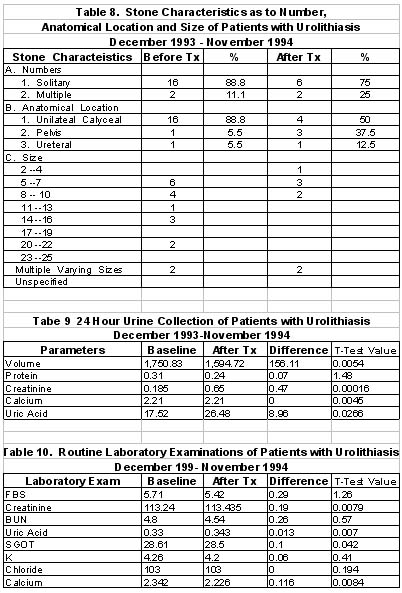
6. Majority of the stones on urograph or sonograms were solitary (78%) and multiple (22%). Most of the stones were located in the upper urinary tract (89%), specifically in the calyces. Of those with solitary stones, the size range from >5 to 10 mm in their greatest axial diameter in 83% and the rest had varying sizes. Radiographically, 90% of the stones were radio-opaque.
7.5.2 Results
Sambong treatment for 6 weeks did not alter significantly the 24 hour urinary
volume, protein, creatinine, calcium or uric acid. Serum uric acid, calcium,
FBS, BUN, creatinine, potassium, chloride and calcium were not affected by
sambong intake.
Clinical monitoring of heart, respiratory rates and blood pressure showed no significant changes during therapy.
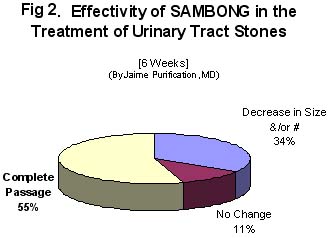
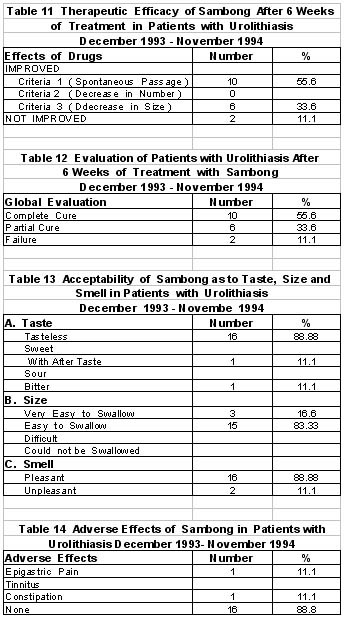
Radiographic evidence of complete stone passage was demonstrated in 10 patients (55.6%) after 6 weeks of sambong intake. In 6 patients (33.6%), decrease in number of stones was shown. Only 2 patients did not show any change radiographically. An over-all radiographic improvement of 89.2% with sambong was documented. Clinical criteria for improvement was seen in 89.2 % of patients, with passage of sandy urine. ( Figure 2)
Adverse effects were complained of by 2 patients (11%) consisting of epigastric pain and constipation.
The sambong tablets were evaluated as acceptable as to taste, smell and ease of swallowing by 88.8% of patients.
7.5.3 Conclusion
1. Urolithiasis among Filipino patients is more common among males than females (1.56:1 ratio) but not to the same extent as among Japanese. The mean age of patients was 37 years with peak at the 3rd and 4th decades, associated with sedentary lifestyle and poor water intake.
2. Stones in the urinary tract usually manifest as back pain or tenderness with accompanying urinary disturbances requiring use of anti - microbials, uricosuric drugs and antispasmodics.
3. Majority of stones on radiography/ sonography were radio-opaque and solitary occurring at the upper urinary tract (calyces).
4. Sambong intake at 40 mg/kg daily for 6 weeks resulted in objective evidence of improvement (radiographically or actual passing out of stones) in 89.2% of patients. Based on the global evaluation, there were 55.6% with complete cure, 33.6% with partial cure and 11.1% failure.
5. Adverse reactions were seen in 2 patients manifested as either epigastric pain or constipation.
6. Acceptability of sambong tablet was evaluated as good by 88.8% of patients.
EXTENDED PHASE III
Open Clinical Trial of Blumea balsamifera, L (Sambong) in the Treatment of Urinary Stones (By: Jaime M. Purificacion, M.D.)
Clinical Abstract
Eighteen patients with urinary stones more than 5 mm in widest diameter seen at the Out Patient Department of the Philippine General Hospital from December 1993 – November 1994 were entered in an open clinical trial to determine the therapeutic efficacy, acceptability as well as to determine the effect of Sambong on renal colic of stone origin and describe the side effects in the treatment of urinary tract stones.
Six weeks were allotted for the treatment period. Improvement, cure and failure were defined.
Results showed that sambong did not significantly alter the 24 hour urinary volume, pH, protein, creatinine, uric acid and calcium as well as serum FBS, BUN, creatinine, potassium, calcium and uric acid.
Radiographic evidences of decrease in size of stones and spontaneous passage of stone in 89.2% of patients was significant.
Sambong tablet is also 88% acceptable with regards to its pleasant odor, taste and easy to swallow tablet preparation.
It would have been ideal if an in-patient setting was done for this study for the purpose of stricter enforcement of accurate 24 hour urine collection since so much of the important parameters that need to be measured depend on urine volume.


BIBLIOGRAPHY
1.
2.
Andrian, R. and
3.
Bagely, D. Pharmacologic Therapy of Urinary Calculi: An Introduction. Urologic
Clinics of
4. De Leon, D. Blumea balsamifera: A Clinical Trial on Its Diuretic Effects. UP-PGH. 1984
5. Finlayson, B. Renal Lithiasis in Review. Urologic Clinics of
6.
Goldwasser, B., Weinerth, J. and
7. Hautea, R., Agulto, M., et al. A Comparison Between Acetazolamide and Blumea balsamifera in Reducing Intraocular Pressure in Primary Glaucoma, UP-PGH, 1990.
8.
Lalli, A. Roentgen Aspects of Renal Calculous Disease. Urologic Clinics of
9.
Lemann, J. Nephrolithiasis. Textbook of Nephrology.
10. Ohkawa, T. and Morimoto, S. Methods for Preventing the Recurrence of Urinary Stone Disease. Asian Medical Journal. 1987; 30(3): 143-148
11. Pak, C., et al. Ambulatory Evaluation of Nephrolithiasis: Classification, Clinical Presentation and Diagnostic Criteria. The American Journal of Medicine. 1980; 69: 19-29
12.
Preminger, G. Pharmacologic Treatment of Calcium Calculi. Urologic Clinics
of
13. Rico, F., et al. Blumea balsamifera: Its Effects on Calcium Stone. EAMC, 1990.
14. Sigua, H., et al. Blumea balsamifera for the Treatment of Urinary Tract Stones: A Randomized Double-Blind Placebo Controlled Study. UP-PGH, 1991.
15. Schwartz, S. Principles of Surgery. Third Edition, MacGraw-Hill Book Co., 1979: 1685-1687.
16.
Smith,L. Medical Evaluation of Urolithiasis: Etiologic Aspects and Diagnostic
Evaluation. Urologic Clinics of
17.
18.
Thomas, W. Medical Aspects of Renal Calculous Disease. Urologic Clinics of
19.
Yachiku, S. and Kiode, T. Drug Therapy for Urolithiasis. Asian Medical Journal.
1987; (3): 156-162.

 |
|
|

|
|
Copyright © 2002 Altermed Corporation. All rights reserved.