|
These educational activities, certified by accredited providers, were
not prepared by Medscape's editors, but are made available on our site
as a service to our audience. Authors are routinely instructed by the
provider to disclose significant financial relationships and mention
of investigational drugs and unapproved indications. Medscape has
received a fee for posting these activities. Direct questions or
comments to: CME@webmd.net.
|
This CME activity, "HRT and SERMs: New Guidelines for Patient Management," was originally offered as a supplement to the March 2002 Special Edition to The Female Patient, certified for CME. The original supplement was published prior to the early termination of the estrogen/progestin arm of the Women's Health Initiative (WHI) trial in July 2002 due to adverse outcomes. The companion estrogen-alone trial continues, with close monitoring by the data and safety monitoring board. The Women's International Study on long Duration Oestrogen after Menopause (WISDOM) trial was halted in October 2002 because the main funding body of WISDOM, the Medical Research Council (UK), withdrew funding for the study. More recent references discussing trial outcomes have been added below, and are recommended reading for completion of this CME activity.
Faculty affiliations and disclosures are at the end of this activity.Release Date: March 31, 2003; Valid for credit through March 31, 2004
Target Audience
This activity was developed for OB/GYNs and primary care physicians.
Goal
Information on estrogen replacement is rapidly becoming available. At the same time, practitioners suggest the information is not always clear. This piece will clarify what is now known about HRT and SERMs, as well as where the field may be heading.
Learning Objectives
Upon completion of this educational activity, participants should be able to:
- Create a treatment plan for the prevention of osteoporosis.
- Describe the effects of cardiovascular disease in postmenopausal women.
- Discuss gynecologic considerations in postmenopausal women.
- Discuss breast cancer and SERMs.
Credits Available
Physicians - up to 2.0 AMA PRA category 1 credit(s)
All other healthcare professionals completing continuing education credit for this activity will be issued a certificate of participation.
Participants should claim only the number of hours actually spent in completing the educational activity.
Canadian physicians please note:
Medscape's CME activities are eligible to be submitted for either Section 2 or Section 4 [when creating a personal learning project] in the Maintenance of Certification program of the Royal College of Physicians and Surgeons, Canada [RCPSC]. For details, go to www.mainport.org.Accreditation Statements
For Physicians
This activity has been planned and implemented in accordance with Essential Areas and Policies of the Accreditation Council for Continuing Medical Education (ACCME) through the joint sponsorship of The Annenberg Center for Health Sciences at Eisenhower and Medscape, Inc. The Annenberg Center is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
The Annenberg Center for Health Sciences designates this educational activity for a maximum of 2.0 hours in category 1 credit toward the AMA Physician's Recognition Award. Each physician should claim only those hours of credit that he/she actually spent in the educational activity.
This activity consists of a website. Successful completion is achieved by reading the material, reflecting on its implications in your practice, and completing the assessment component.
The estimated time to complete the activity is 2 hours.
For questions regarding the content of this activity, contact the accredited provider for this CME/CE activity: mpederson@annenberg.net. For technical assistance, contact CME@webmd.net.
Instructions for Participation and Credit
This online, self-study activity is formatted to include text, graphics, and may include other multi-media features.
Participation in this self-study activity should be completed in approximately 2.0 hours. To successfully complete this activity and receive credit, participants must follow these steps during the period from March 31, 2003 through March 31, 2004.
- Make sure you have provided your professional degree in your profile. Your degree is required in order for you to be issued the appropriate credit. If you haven't, click here. For information on applicability and acceptance of continuing education credit for this activity, please consult your professional licensing board.
- Read the target audience, learning objectives, and author disclosures.
- Study the educational activity online or printed out.
- Read, complete, and submit answers to the post test questions and evaluation questions online. Participants must receive a test score of at least 70%, and respond to all evaluation questions to receive a certificate.
- When finished, click "submit."
- After submitting the activity evaluation, you may access your online certificate by selecting "View/Print Certificate" on the screen. You may print the certificate, but you cannot alter the certificate. Your credits will be tallied in the CME Tracker.
The time for this program has never been better: Researchers are intensely exploring the role of female hormones in the development of age related illnesses such as cardiovascular disease, osteoporosis, and cancer. Such investigation has unveiled estrogen's benefits in preserving bone mineral density (BMD) and preventing certain gynecologic cancers, and has provided evidence that combined estrogen and progesterone exert positive effects on serum lipid levels and, quite possibly, the risk of coronary artery disease. This investigation has also unveiled much controversy, which has complicated prevention and treatment protocols.
As diligent as many healthcare providers have been in communicating to patients about the various advantages of estrogen replacement therapy (ERT) and hormone replacement therapy (HRT), many postmenopausal women have been less enthusiastic about long-term use of these interventions. Although the many benefits of supplemental estrogen have been documented in numerous studies, several factors contribute to many patients' lack of interest in and compliance with ERT and HRT. These include real and perceived breast tenderness, weight gain, and skin changes, as well as the possible risks of breast and ovarian cancer.
Because of these limitations on ERT and HRT, a great emphasis has been placed on identifying estrogen-related therapies that postmenopausal women might perceive as less fraught with side effects and risks. These efforts have yielded selective estrogen receptor modulators (SERMs): compounds that act efficiently through the estrogen receptor but do not increase the amount of active hormone in the body. The first agent observed to act in this manner was tamoxifen, used for the prevention of breast cancer in women at high risk for the disease. Tamoxifen has also been widely observed to exert beneficial effects on bone,[1] and, more recently, markers for cardiovascular health.[2] However, tamoxifen has also been shown to incur some risk of ocular disorders and uterine cancer.[3,4]
Further efforts to improve upon the prevention of age-related disease in postmenopausal women have led to the development of another SERM, raloxifene, designed for even higher selectivity in this population. Raloxifene is currently under investigation for the prevention of breast cancer and may actually hold promise in several therapeutic areas. Readers should note, however, that the risk of thromboembolism is the same in women using raloxifene as in those using exogenous estrogen, and identification of appropriate disease markers in at-risk patients is warranted. The areas in which SERMs are being explored for optimum benefit were discussed in a roundtable meeting entitled "HRT and SERMs: Evidence-Based Data to Guide Patient Management," the proceeds of which are presented here in the form of clinical articles and an additional dialogue section titled "The Faculty Speaks." Brief summaries of the clinical articles appear below.
Raloxifene decreases bone turnover in a manner similar to that seen with estrogen replacement. As discussed in the article, "Osteoporosis: Fractures and Key Risk Factors" by Robin K. Dore, MD, this agent has been extensively studied for its use in reducing osteoporotic fracture in the spine, hip, and total body.
As cardiovascular disease (CVD) is the number-one killer of women (and men), healthcare providers are generally enthusiastic about the discovery of cardiovascular benefits in a new drug. Currently, there is good reason to believe that the SERMs offer some consistent and positive effects on low-density lipoprotein cholesterol, without increasing serum triglycerides or decreasing high-density lipoprotein cholesterol.[2] The article, "Cardiovascular Disease: New
Recommendations for Minimizing the Threat," by Lori Mosca, MD, MPH, PhD, provides thoughtful insight into these and other therapies to reduce the incidence of CVD among older women.
SERMs represent a very exciting option for disease prevention in certain postmenopausal women. In enthusiastic response to our readers, the articles in this program aim to "cut through the noise" that too often clouds the issue of postmenopausal hormone-related therapy. It is my hope that you, the reader, will be able to draw from the insights published here to enhance the overall well-being of our postmenopausal patient population.
Breast cancer is the number-one cause of new cancers in women. An estimated 192,200 new cases were diagnosed in 2001.[1] Breast cancer is the number two cause of death from cancer in women. Whereas approximately 25% of cancer deaths in women are due to cancer of the lung and bronchus, 15% are due to breast cancer, and it is estimated that about 40,000 women may have died in 2001 from this disease.[1]
Most clinicians are very familiar with the risk factors for breast cancer (Table 1). Recently, there have been significant findings regarding risk factors for breast cancer, however, which are worthy of discussion.

Table 1. Risk Factors for Breast Cancer in Women
In 1997, the Collaborative Group on Hormonal Factors in Breast Cancer published their reanalysis of data from 51 epidemiologic studies, which included approximately 161,000 women in 21 countries, including almost 53,000 women with breast cancer and more than 108,000 without the disease.[2] This meta-analysis showed an approximate 2% increase in the risk of breast cancer per year of use of estrogen replacement therapy (ERT). A woman who used estrogen for longer than 15 years had a 60% increase in relative risk for developing breast cancer. This risk was particularly evident among lean women with lower body mass index (BMI). The meta-analysis also found that the increase in risk disappeared almost completely 5 years after cessation of ERT.
A number of methodologic challenges and limitations have been identified in regards to the Collaborative Group study. One is that it primarily focused on ERT alone, with a very small number of patients receiving a combination of estrogen and progestin (HRT). A cohort study of followup data from the Breast Cancer Detection Demonstration Project was undertaken to determine whether the risk associated with combination HRT was greater than the risk associated with estrogen alone.[3] In 2000, results were published suggesting that the combination regimen increased risk beyond that associated with estrogen alone (Table 2).
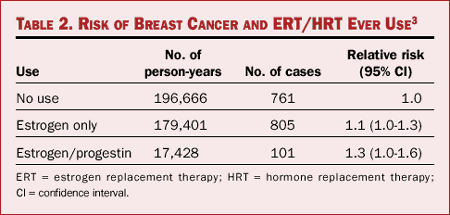
Table 2. Risk of Breast Cancer and ERT/HRT Ever Use[3]
Results among leaner women in this study were consistent with the results from the Collaborative Group's meta-analysis. Women with a BMI of 24.4 kg/m2 or lower had an increased risk of breast cancer associated with HRT that was 2.0 (1.3-3.0) at 4 or more years' duration of therapy, compared with 1.3 (0.7-2.4) for women with a BMI of at least 25 kg/m2. The increase in risk was evident for the majority of invasive tumors with ductal histology and regardless of the extent of invasive disease.
Women who are currently entering menopause have been, or have the potential to have been, exposed to oral contraceptives (OCs), and therefore estrogen, for a much longer period than were members of the previous generation. Therefore, it's important to look at how this combination of OC exposure during the premenopausal years and exogenous estrogen starting in the postmenopausal years will affect their risk for breast cancer.
Brinton and colleagues conducted a case control study of breast cancer among 1031 women (919 of whom were controls) under age 55. Results showed that women who had used OCs for 10 or more years and used HRT for 4 or more years appeared to have an increased risk for breast cancer.[4] These results should be interpreted cautiously, given the small numbers within the subgroups, but they do raise a possibility and emphasize the need for further evaluation of breast cancer risk for women considering hormone replacement after many years of exposure to OCs.
Today, after 50 years of use, no randomized trials exist to provide definitive answers about the relationship between ERT/HRT and increased risk for breast cancer, but the studies discussed here raise serious questions. In addition, the well-publicized controversy surrounding ERT/HRT is of great concern to patients. Hopefully, large-scale, controlled studies, such as the Women's Health Initiative Study Group, will begin to provide the answers that are needed.
Circulating levels of endogenous estrogen have been demonstrated to influence a woman's risk for developing breast cancer.[5, 6] Several prospective studies have consistently indicated that women with the highest levels of estradiol appear to have a twofold to threefold excess risk of breast cancer compared with women with the lowest levels of estradiol.[7] These results are important, in that the level of estradiol designated as "high" in these studies was previously considered unremarkable. Therefore, researchers are finding a relationship between estradiol and breast cancer at levels that were once thought to be clinically normal.
Studies that measure estradiol levels from the circulating blood stream rely on a single blood draw, which measures the levels at that moment in time but tells little about a woman's estradiol levels in the longer term. To resolve that limitation, the Study of Osteoporotic Fractures (SOF) Research Group conducted a study of bone mineral density (BMD) and risk of breast cancer based on the hypothesis that, because it contains estrogen receptors, bone can serve as a marker for a woman's lifetime exposure to estrogen.
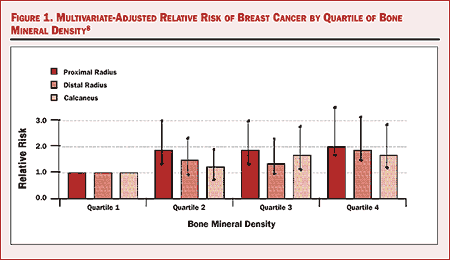
Figure 1. (click to enlarge) Multivariate-adjusted relative risk of breast cancer by quartile of bone mineral density[8]
The group found that BMD did in fact predict the risk of breast cancer in older women (Figure 1).[8] These findings were confirmed in the Framingham Study, which also found that women in the highest quartile of bone mass are at higher risk for postmenopausal breast cancer.[9] In a more recent followup of the SOF prospective study of almost 9000 women older than 65, the relationship between BMD and breast cancer was particularly evident for the more advanced stages of breast cancer.[10]
The mammographic density of breast tissue varies from woman to woman, depending on the relative amounts of fat (which is radiologically clear) and connective and epithelial tissue (which is radiologically dense) that are present. These variations are referred to as the parenchymal pattern of the breast. A premenopausal woman has higher breast density than postmenopausal women, because breast density decreases with age.
Several studies have shown that breast density is related to breast cancer. For example, the Canadian National Breast Cancer Screening study found that women who had more than 75% of their breast made up of high density tissue had a fivefold greater risk of breast cancer compared with women who had less dense breasts (Figure 2).[11]
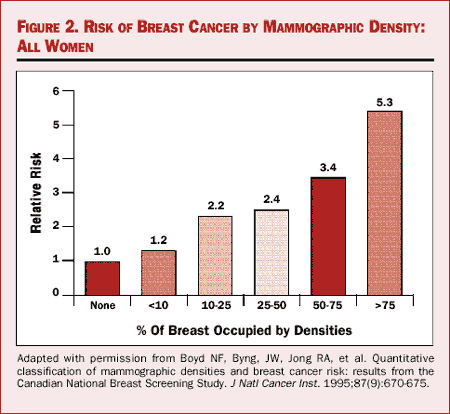
Figure 2. Risk of breast cancer by mammographic density: All women
HRT and breast density: Investigators participating in the Postmenopausal Estrogen/Progestin Interventions (PEPI) trial looked at the effects of hormone replacement therapy on the parenchymal pattern of the breast.[12] Their results showed that the estrogen/progestin combination regimens produced a much greater increase in breast density (from 16.4% to 23.5%) than did estrogen alone (3.5%) or placebo (0%).
A recently published observational study involving more than 5000 women who were followed for more than 2 years showed that the relationship between HRT and breast density is dynamic.[13] Women who initiated HRT over the 2-year period of the study were twice as likely to show increases in breast density, which were sustained while therapy with HRT continued. Breast density decreased with the discontinuation of HRT.
Raloxifene and breast density: To determine the effect that selective estrogen receptor modulators (SERMs) have on breast density, Freedman and colleagues assessed changes in mammographic density among healthy postmenopausal women participating in an osteoporosis prevention trial.[14] The women were randomly assigned to receive raloxifene, unopposed estrogen, or placebo, and were followed for 2 years (Table 3).

Table 3. Effect of Raloxifene and ERT on Breast Density: Study Design[14]
The women who received placebo showed a significant decrease in breast density, as did the women who received raloxifene. The women who received estrogen, on the other hand, experienced a nonsignificant increase in breast density (Figure 3).
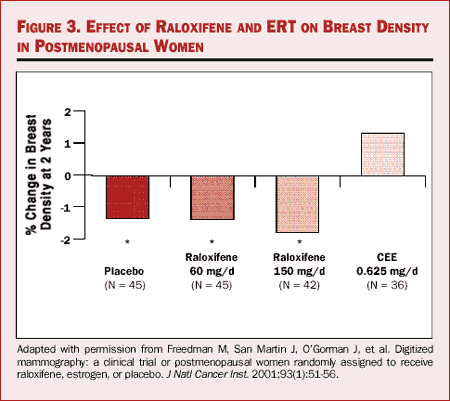
Figure 3. Effect of raloxifene and ERT on breast density in postmenopausal women
Breast cancer risk reduction: Breast cancer was a prespecified secondary endpoint of the Multiple Outcomes of Raloxifene Evaluation (MORE) trial, a randomized clinical trial in postmenopausal women with osteoporosis. A total of 7705 women were enrolled in the 4-year trial, randomly assigned to raloxifene 60 mg/day, raloxifene 120 mg/day, or placebo.[15] Results showed that raloxifene reduced the risk of newly diagnosed invasive breast cancer by 72% in these women. The risk of estrogen receptor-positive invasive breast cancer was reduced by 84% (95% confidence interval, 70% to 90%).
Pooled data from the raloxifene group (5129 subjects, 22 breast cancer cases, 16,958 women-years of follow-up) demonstrated a breast cancer rate per 1000 patient-years of 1.3; the placebo group (2756 subjects, 39 breast cancer cases, 8,356 women-years of follow-up) demonstrated a rate of 4.7.[15]To test the hypothesis that risk factors related to lifetime estrogen exposure predict breast cancer incidence, and to find out if any subgroups experienced enhanced benefits from raloxifene, MORE investigators analyzed breast cancer risk by the following baseline characteristics indicative of estrogen exposure: baseline serum estradiol, BMI, femoral-neck BMD, prevalent vertebral fractures, previous HRT use, and family history of breast cancer.[16] Results confirmed that a higher lifetime estrogen exposure increases breast cancer risk. Women with the highest third of estradiol levels ( ≥ 12 pmol/L) had a 2.07-fold increased risk of developing invasive breast cancer compared with women who had lower levels. Raloxifene significantly reduced the risk of breast cancer in postmenopausal osteoporotic women, regardless of lifetime estrogen exposure. The reduction was greater in patients with higher lifetime estrogen exposure.
Tamoxifen and breast cancer prevention: Three studies have looked at the effect of tamoxifen in breast cancer prevention. The largest was the Breast Cancer Prevention Trial (BCPT), initiated in 1992.[17] This trial enrolled more than 13,300 women in the United States and Canada. One of the criteria for participation in the trial was a 5-year predicted risk for breast cancer of at least 1.66% or a history of lobular carcinoma in situ. In other words, the women who participated in the BCPT were required to be at high risk. Tamoxifen reduced the risk of noninvasive beast cancer by 50%. It also reduced the occurrence of estrogen receptor-positive tumors by 69%; however, it had no effect on the occurrence of estrogen receptor-negative tumors. This trial led to the approval by the US Food and Drug Administration of tamoxifen for the prevention of breast cancer among patients at high risk.
During the same year that the BCPT trial was published (1998), two other large, randomized clinical trials evaluating tamoxifen in the prevention of breast cancer were also published. One was an interim analysis of a study being conducted by investigators at the Royal Marsden Hospital in London.[18] This trial enrolled nearly 2500 healthy women with a family history of breast cancer and randomized them to receive either tamoxifen 20 mg/per day or placebo for up to 8 years. The interim analysis did not show any effect of tamoxifen on breast cancer incidence among the women in this trial. Investigators theorized that differences in study populations between this trial and the BCPT could account for the lack of efficacy: Eligibility for the Royal Marsden trial was based predominantly on a strong family history, whereas eligibility for the BCPT was based mostly on nongenetic factors.
The second trial, the Italian Tamoxifen Prevention Study, was a placebo-controlled study of tamoxifen in more than 5000 women who did not have breast cancer and who had undergone hysterectomy.[19] Investigators restricted eligibility to women who had been hysterectomized, out of concern about the observation that tamoxifen increases the risk of endometrial cancer. In fact, a large proportion of the women in this trial had undergone a bilateral hysterectomy. Results of the trial showed no difference in breast cancer frequency between the placebo (22 cases) and tamoxifen (19 cases) arms.
The possibility of significant breast cancer prevention with SERMs appears promising. Data from the MORE trial in women with osteoporosis showed that at 4 years, raloxifene produced a highly significant reduction (72%) in the risk of newly diagnosed invasive breast cancer, with an even greater reduction (84%) in the risk of estrogen receptor-positive invasive breast cancer. In addition, raloxifene significantly reduced breast cancer risk in these women regardless of their lifetime estrogen exposure.
The BCPT showed that tamoxifen reduced the risk of noninvasive beast cancer by 50% and the occurrence of estrogen receptor-positive tumors by 69%, leading to its approval for breast cancer prevention in women at high risk. The Royal Marsden and Italian studies resulted in no significant reductions in breast cancer risk with tamoxifen, but variations in eligibility between these trials and the BCPT trial may account for the differences in outcome.
The following ongoing trials will provide more definitive answers about the role of HRT and SERMs in the prevention of breast cancer and the role of ERT/HRT in risk for breast cancer:
Although much of these data will not be available for several years, preliminary results from one or more of these trials may begin to provide the important answers that clinicians and patients are awaiting.
Postmenopausal health concerns, including age related cancer risks, are rapidly becoming a larger public health priority. More and more women are entering their postmenopausal years, driving research efforts further in uncovering the benefits and risks of both standard and evolving therapies for postmenopausal conditions. As a result, long-standing beliefs regarding traditional hormonal therapies are being rethought; accumulating evidence for newer drug treatments is reshaping the therapeutic landscape of postmenopausal health.
Prescription therapies for postmenopausal symptoms are predominantly hormonal; however, newer agents are gaining ground. Selective estrogen receptor modulators (SERMs) in particular are receiving increasing attention as an alternative to estrogen replacement therapy (ERT). Although significant research remains to determine the full scope of benefits and risks associated with SERMs, findings to date have provided important information on how these compounds affect the gynecologic changes associated with postmenopause.
As with any drug therapy, there are risks involved and concerns that must be considered before treatment is initiated and, in many cases, followed throughout treatment. In the case of hormone therapy, research continues to provide new and useful insights into its effects on postmenopausal health. Estrogen continues to be the "gold standard" for the treatment of vasomotor symptoms and vaginal atrophy. However, the understanding of estrogen's effects on other gynecologic problems continues to evolve.
It is well established that unopposed estrogen, or ERT, is associated with an increased risk of endometrial cancer.[1-3] Studies have indicated that the risk of developing endometrial cancer increases with duration of ERT use,[4-7] and that the risk may persist for more than 10 years after discontinuation of just 1 year of ERT.[8] The addition of progestin largely reduces the risk for endometrial cancer induced by estrogen, likely by reducing estrogen receptor activity.[9] In the Postmenopausal Estrogen/Progestin Intervention (PEPI) trial, 10 women taking unopposed estrogen during the 3-year study had a significantly increased risk for endometrial hyperplasia compared with those taking estrogen plus progestin (34% vs 1%).
Today, the administration of estrogen plus progestin, termed hormone replacement therapy (HRT), is standard therapy for women who still have a uterus. However, the question remains as to whether cyclic or continuous combination therapy offers the same level of protection against endometrial cancer. To date, clinical trials have shown continuous combination therapy to offer significantly greater protection against endometrial cancer compared with cyclic therapy. After 15 years of follow-up, Wiederpass and colleagues noted a dramatic difference between the two regimens, with a relative risk of 3.5 in women taking cyclic therapy, compared with a relative risk of 0.2 in those taking continuous combination therapy.[11] This and other confirmatory findings indicate that for long-term use, continuous combination therapy is preferable for optimal protection against endometrial cancer. Regardless of which treatment regimen is used, monitoring for endometrial cancer should be performed regularly.
The North American Menopause Society has outlined screening parameters for endometrial cancer for women who are administered cyclic and continuous HRT. With cyclic HRT, a baseline pelvic examination should be performed, with endometrial evaluation considered for bleeding other than withdrawal bleeding, or for heavier or prolonged withdrawal bleeding. Baseline pelvic examination should also be performed for women administered continuous HRT, with endometrial evaluation considered if irregular bleeding occurs more than 6 months after starting therapy. Early biopsy can be considered on a case basis. Women with a uterus generally should not take unopposed estrogen, but if administered, baseline and annual routine pelvic and endometrial evaluations are recommended.[27]There has been a long-standing belief among gynecologists that estrogen therapy is beneficial for urinary incontinence. However, studies conducted over the last few years have found that estrogen therapy does not improve incontinence, and in fact, may actually make the condition worse.
Recent data concerning the effects of estrogen on urinary incontinence come from the Heart and Estrogen/Progestin Replace Study (HERS), a randomized, blinded trial of the effect of hormone therapy on recurrent coronary heart disease events among 2763 postmenopausal women younger than 80 years with coronary disease and intact uteri.[12] Study participants were randomly assigned to HRT or placebo and followed for an average of 4.1 years. In a post-hoc analysis, the severity of incontinence at the end of the trial was evaluated for the subgroup of the cohort who had reported at least one episode of incontinence per week at the baseline evaluation. Severity of incontinence was classified as improved (if there was a decrease of at least two episodes per week), unchanged, and worsened (if there was an increase of at least two episodes per week).
The study found that incontinence improved in more women taking placebo than those taking HRT. Moreover, for women whose condition worsened, those taking HRT actually experienced more problems than those who were taking placebo. Thus, the results indicated that daily oral HRT was associated with worsening urinary incontinence in older postmenopausal women with weekly incontinence and that HRT should not be a recommended treatment. These findings warrant closer examination of estrogen and its influence on urinary incontinence.
Raloxifene and tamoxifen are the two primary SERMs currently prescribed as alternatives to hormone therapy in menopausal and postmenopausal women. Raloxifene (Evista) is indicated for the prevention and treatment of osteoporosis, and tamoxifen (Nolvadex) is indicated for the treatment of breast cancer and to reduce the risk of breast cancer in high-risk women. In addition to these primary indications, raloxifene and tamoxifen both have demonstrated other beneficial effects as well as risks related to postmenopausal health.
Clinical trials have found a clear association between tamoxifen and endometrial cancer. In the Breast Cancer Prevention Trial (BCPT),[13] which involved 13,388 premenopausal and postmenopausal women, tamoxifen dramatically reduced the risk of breast cancer by 49%. However, the risk of invasive endometrial cancer was 2.5 times greater with tamoxifen. These findings of significant benefit tempered by serious risk have been verified in other trials of tamoxifen,[14, 15] and must be weighed when tamoxifen is being considered. For many women, the danger of endometrial cancer is not so great a threat compared with the decrease in breast cancer risk promised by tamoxifen, particularly as it appears that most of these endometrial cancers are well-differentiated tumors. In any case, there is a definite risk of endometrial cancer with tamoxifen, and every woman taking tamoxifen should be monitored. Recommendations from the American College of Obstetricians and Gynecologists for monitoring women on tamoxifen are outlined in the table below.

Table. Recommendations for Monitoring Women Taking Tamoxifen
Preclinical data from animal models indicates that raloxifene does not stimulate the endometrium, nor does it increase uterine thickness, which is often used as a risk marker.[16] More recent evidence from human trials supports the preclinical data. In these reports (following 7700 patients), endometrial thickness was unchanged during raloxifene therapy at doses up to 150 mg/d, compared with placebo.[17, 18] In the Multiple Outcomes of Raloxifene Evaluation (MORE), raloxifene did not increase the risk of endometrial cancer.[19] After 40 months of follow-up, the rate of endometrial cancer was slightly lower in women receiving raloxifene, although not statistically significant. In the latest report from MORE, which comes after 48 months of follow-up, raloxifene continues to maintain a slightly lower rate of endometrial cancer compared with placebo.[20]
Although further assessment is needed to determine the clinical influence of raloxifene on endometrial cancer, findings to date suggest that raloxifene, unlike tamoxifen, does not have an adverse effect on the endometrium. Whether it will ultimately prove to have a protective effect remains to be seen.
Data is very limited concerning the effects of tamoxifen on ovarian cancer. Tamoxifen has been shown to stimulate ovarian cysts in premenopausal women, however, studies to date have not demonstrated any increased risk for ovarian cancer in women taking long-term tamoxifen treatment.
Even less is known about the effects of raloxifene on ovarian cancer. Recent laboratory tests in rodents showed that raloxifene stimulated the growth rate of ovarian cancer cells.[21] However, of the human clinical trials of raloxifene conducted thus far, none have noted an increase in ovarian cancer.
A recent assessment of safety data from three clinical trials of raloxifene found that raloxifene does not increase pelvic floor relaxation.[22] For this evaluation, data was collected from nearly 7000 women taking either raloxifene or placebo on the number of surgeries that had been performed either for prolapse or urinary incontinence.
The investigators found that there were fewer surgeries for prolapse among women taking raloxifene, and concluded that raloxifene significantly reduced the risk for pelvic floor surgery. This evaluation is limited in that it was a post-hoc analysis and prolapse was not an objective in the three trials analyzed. As such, it cannot be presumed that raloxifene reduces the risk of prolapse; however, it can be assumed that, unlike idoxifene and lasofoxifene, raloxifene does not cause an increase in the risk of pelvic floor relaxation or prolapse.
New findings from an osteoporosis prevention study comparing raloxifene with estrogen therapy have shown that raloxifene was not associated with an increased risk for urinary incontinence.[23] The study included 619 postmenopausal women who were randomly assigned to estrogen, raloxifene, or placebo. The prevalence of urinary incontinence at baseline was similar across all groups.
After 3 years, there was no statistically significant difference in the incidence of urinary incontinence among the placebo and raloxifene groups. New or worsened episodes were reported by 1.3% of the placebo group and 0.7% of the raloxifene group. In contrast, 7% of the estrogen group reported new or worsened episodes of urinary incontinence, representing a significant increase. Moreover, of all study participants reporting urinary incontinence, 80% of those taking estrogen rated the severity of their symptoms as moderate to severe, while those receiving raloxifene rated the severity of their symptoms as mild.
These findings are consistent with those from the HERS trial[12], which suggested that estrogen does not appear to prevent urinary incontinence, but instead may increase a woman's risk of developing the condition.
Symptomatic patients generally find relief from hormone interventions, whereas asymptomatic patients often report that hormone therapy adversely affects their quality of life.[24] Based on an analysis of eight small randomized trials of raloxifene and the MORE trial, it appears that raloxifene is fairly well tolerated. In contrast to estrogen and estrogen plus progestins, raloxifene does not cause breast tenderness or uterine bleeding.[24, 25] In clinical trials, 4% to 7% more women taking raloxifene compared with those taking placebo report hot flushes,[25, 26] but these flushes are usually mild and seem to resolve after a few months. Compared with placebo, raloxifene is also associated with flu-like symptoms and leg cramps, but these adverse effects are uncommon and rarely lead to discontinuation of the medication.[25, 26]
The most data on menopausal symptoms associated with tamoxifen are from the large BCPT study.[13] Based on the results of this trial, the side effects experienced most often by women taking tamoxifen were vaginal discharge and hot flashes. Hot flashes of any severity occurred in 68% of women on placebo and in 80% of women on tamoxifen. Vaginal discharge occurred in 35% and 55% of women on placebo and tamoxifen, respectively, and was severe in 4.5% and 12.3%, respectively. Women in both groups reported side effects such as vaginal dryness, itching, or bleeding; menstrual irregularities; depression; appetite loss; and headaches.
As traditional assumptions about hormone therapy continue to be challenged by new, more rigorous research, it is imperative to gain a better understanding of other treatment options for postmenopausal health conditions. The SERMs raloxifene and tamoxifen are two of these options. A large number of postmenopausal women are entering new clinical trials or are being followed in ongoing trials evaluating the effects of tamoxifen and raloxifene on a wide range of clinical endpoints. Findings from this research may influence future recommendations regarding these SERMs, and warrant ongoing attention.
The following are excerpts from a closed roundtable discussion that took place in October, 2001, entitled "HRT and SERMs: Evidence-Based Data to Guide Patient Management." Dr Thomas E. Nolan acted as moderator.
As noted in "Cardiovascular Disease: New Recommendations for Minimizing the Threat," by Lori Mosca, MD, MPH, PhD, a great need exists for education, screening, and intervention for cardiovascular illness in postmenopausal patients.
Dr Nolan: When it comes to the prevention and management of cardiovascular disease (CVD), for many years we've segregated male and female patients. However, data are beginning to show that women have some of the same problems with coronary arteries as men do. Also, many OB/GYNs are not well attuned to the difference between primary and secondary prevention; in many cases, by the time secondary prevention is needed, they have already lost control of that patient.
Dr Johnson: What should be the approach in treating women who don't have proven coronary heart disease but are at very high risk, because of such conditions as diabetes and hyperlipidemia? Should we be giving them estrogen?
Dr Mosca: The question you are raising is one of risk equivalence. The American Heart Association guidelines do not recommend estrogen for secondary prevention. For nearly all postmenopausal women, there's some degree of atherosclerosis. At some point, when it becomes clinically evident, we call its management "secondary prevention." The problem with that way of thinking is that the first time a woman manifests coronary heart disease, two thirds of the time it's with sudden death. There are no prior symptoms. The OB/GYN is in a unique position to treat women more aggressively and to not wait until there is a diagnosis.
Even in women who are 55 or recently postmenopausal and have one or more risk factors, I am aggressive in the prevention of CVD.
Dr Nolan: I try to intervene with patients early in menopause as well. I'm not always successful, because patients are often not yet willing to accept menopause-related interventions.
Dr Mosca: As for hormone replacement therapy (HRT), it's generally not being used now as a mechanism for the prevention of heart disease. In the cardiovascular community, the data appear to be insufficient to use it when there are many other methods [eg, physical activity, smoking cessation, nutrition, lipid lowering via statin therapy, blood pressure medication] of controlling CVD.
Dr Dore: What about primary versus secondary prevention and low dose aspirin in women?
Dr Mosca: For secondary prevention, aspirin is well accepted. For primary prevention, there's an ongoing trial right now. We do not generally recommend aspirin in the setting of primary prevention.
Even though a benefit has been seen on an epidemiological level, mainly among women with risk factors, aspirin therapy may increase risk of hemorrhagic stroke.[1] Stroke accounts for nearly one third of all cardiovascular events in women. Therefore, until we really have an evidence base to suggest that aspirin lowers overall cardiovascular mortality, there isn't a lot of enthusiasm to use it right now.
Dr Cauley: The Women's Health Initiative and the Heart and Estrogen /Progesterone Replacement (HERS) studies both use conjugated equine estrogen (CEE) and medroxyprogesterone acetate (MPA). What about some of the other forms of HRT? My understanding is that they're now being used more frequently.
Dr Mosca: The world literature still largely pertains to CEE and MPA. One school of thought exists that different estrogens and progestins may exert different cardiovascular effects. In particular, there is a lot of enthusiasm in Europe over 17-beta estradiol. Last year, Hodis presented an estradiol study showing a reduction in the progression of corroded intima-media thickness.[2]
In terms of progestins, there is a lot of controversy surrounding whether or not one progestin might be better than another. The literature is quite mixed. The take-home message is that we don't know whether or not one form of estrogen and/or progestin is better. It's worth pursuing these studies to see if we can develop a database that qualifies for a larger-scale critical outcome study.
Unfortunately, the long-term epidemiologic studies of secondary prevention have been minimal. So, in this case, epidemiology data seem to be consistent with the randomized trials. And there is no primary prevention outcome study yet.
Dr Nolan: I see a lot of symptomatic depression in postmenopausal women, which is also a considerable risk factor.
Dr Mosca: Indeed. Although the Enhancing Recovery in Coronary Heart Disease [ENRICHD] trial, the first major randomized, clinical trial to test treating depression and low social support with psychosocial intervention, did not show a reduction in cardiovascular endpoints, there was a statistical improvement in depression and social isolation.[3] There is great opportunity for us to make sure that women get appropriate treatment for these problems. I suspect that depression, anxiety, and social isolation are major contributors to CVD for women.
Dr Nolan: Our community is now really looking for depression and treating it. Dr Johnson, you and I were talking about using selective serotonin reuptake inhibitors (SSRIs) for chronic pelvic pain and premenstrual dysmorphic disorder several years ago.
Dr Johnson: Yes, and at present, a lot more practitioners are now prescribing SSRIs for these and other hormone-related psychological mood disorders.
Dr Dore: Thanks to the higher level of social acceptance than these agents once had, patients as well as clinicians are now more willing to discuss and use these therapies.
Dr Nolan: Indeed, and over the years, I'm finding less and less reluctance. Twenty years ago, a colleague of mine said CVD was related to chronic stress causing damage to the arterial walls. I thought it was an interesting theory, and I still think there may be an element in there somewhere.
Dr Mosca: Absolutely. The role of psychosocial markers in CVD is now very prominent. A number of studies have examined, for example, type A personality as a risk factor for cardiovascular events. Unfortunately, most of this research has been conducted in men. Only in recent years have even the major studies begun to collect information about how personality traits and reactions to environmental stress affect heart health. We all suspect that these factors are major contributors to CVD in women.
Another issue is the multiplicity of the family and societal roles women now have. For many years, women have been the caretakers of their spouse and children; now, they are often also in the "sandwich role" of caregiver for a parent, while also working outside the home. How the psychosocial environment contributes to CVD remains to be determined. Common sense tells us that it doesn't bode well to have competing priorities. Because of these responsibilities, many women tell us that they don't have time to exercise or to go to cardiac rehabilitation, for example. They feel their need to care for others is greater than that to care for themselves.
When the World Trade Center disaster occurred, we set up a heart attack prevention center downtown. We did it because severe emotional stress leads to a host of responses from the body: Viscosity is increased, fibrinogen levels are increased, blood pressure is increased, and cholesterol is even increased in some studies. All of these markers can acutely affect risk among individuals who have unstable plaque.
Here is another example: There tends to be about an eight fold increase in the risk of cardiovascular events in the 2 weeks following earthquakes and other natural disasters. For about 6 months, cardiovascular mortality rates remain doubled.
Dr Dore: Nutraceuticals are very important to my patients. Have any trials looked at soy or red clover?
Dr Mosca: The soy data have been mixed, also. Some surrogate endpoint studies, such as those on endothelial function, have been positive.[4] Others have been neutral.[5] Many women like the idea of using a natural form of estrogen rather than a pharmaceutical compound. I have concerns about that, because I don't know exactly what they're getting or what it's doing to their blood vessels. There's evidence that soy, when it's replacing animal protein, can lower cholesterol.[4] Again, whether these intermediate endpoint data are going to translate into beneficial clinical outcomes remains to be determined. It would be very nice if we could see a well-conducted trial of soy in postmenopausal women. In terms of our recommendations, it's premature right now to say that soy has a cardiovascular benefit.
Awareness is the key to preventing osteoporosis. Here, Robin Dore, MD, author of "Osteoporosis: Focusing on Fracture and Key Risk Factors," discusses this disease with the other faculty
members, offering critical information on monitoring, prevention, and treatment.
Dr Dore: My primary goal in speaking to patients and physicians is to increase their awareness of osteoporosis prior to the fracture so we can prevent those fractures from occurring. Luckily, we have many therapies for preventing and/or treating these patients, including alendronate, raloxifene, risedronate, and calcitonin. Estrogens no longer have the indication for the treatment of osteoporosis as there has been no clinical evidence indicating that estrogen reduces the risk of fractures. In the future, maybe next year, we will have therapies that are anabolic and actually increase bone formation. So what we can do today is identify the patients who are at risk, start them on therapy, and have them be ambassadors of bone health.
We need to constantly emphasize to adolescents the importance of building peak bone density -- through calcium intake, weightbearing exercise, and not smoking.
Dr Cauley: The National Osteoporosis Society guidelines do not recommend follow-up bone mineral density (BMD) measurement once a woman is on therapy. Why wouldn't you need them?
Dr Dore: The National Osteoporosis Foundation guidelines on vertebral fractures assume that the patient has osteoporosis. But a malignancy [eg, multiple myeloma, primary malignancies of bone] can cause those vertebral fractures as well. A patient could have a trauma as a child and not remember having a vertebral fracture.
One of my patients had a vertebral compression fracture at T10. She was on long-term thyroid replacement, had never taken estrogen, was at risk of falling, and had a positive family history of osteoporosis. If I had treated her, she would have taken therapy needlessly, because she had a normal BMD. As it turns out, she had fallen down a flight of stairs when she was 12 -- that was when the fracture occurred.
Dr Cauley: Bone mineral density is a good screening tool. It identifies people at risk for fracture. But it's not perfect. A patient with a BMD higher than -2.5 SD can still have a hip fracture.
Dr Dore: Indeed, the problem with the World Health Organization criteria for osteoporosis is that age and prior fracture are not taken into account. I put age first, fracture history second, and then T-score. So, if a person has a T-score of -1.8 but has a hip fracture, I would certainly treat her. I advise that these guidelines not be used too strictly.
Unfortunately, I've had many patients referred to me for treatment of osteoporosis because they've had a fracture, and it turns out to be a malignancy. I always evaluate a patient for malignancy if she's had a fracture and the assumption has been that it's osteoporotic.
Dr Johnson: Do we know what the prevalence is of morphometric fractures in women based on their BMD? For example, in women with BMD above -2.5 SD and who are 70 years old?
Dr Dore: In 80-year-old patients, the prevalence is 44%.[6] With regard to BMD measurements, a great disparity exists among different machine manufacturers. Therefore, I always recommend using the same machine every time if possible. If that's not possible, the next best thing is to use a machine from the same manufacturer.
Dr Nolan: What about hyperthyroidism and overuse of thyroid replacements?
Dr Dore: Three abstracts at the World Congress of Osteoporosis last year looked at these data. They showed that between 4 and 9 years of having a normal ultrasensitive thyroid-stimulating hormone level, some bone loss occurred. Other recent data showed that one of the greatest risks for hip fractures was longterm thyroid replacement.[7] Thus, it appears that the endogenous thyroid hormone acts differently than taking either exogenous thyroid or exogenous corticosteroids in this regard.
The issue of frozen bone also came up, surrounding the fact that there was increased
tetracycline labeling along the surface of the bone in patients who were taking both alendronate and estrogen. This was not the case in patients taking residronate and estrogen. Also, alendronate has a half-life of about 10 years in bone and residronate is about 400 days.
Any discussion of postmenopausal health interventions warrants the inclusion of their respective safety issues. In this section, the participants focus on the use of SERMs and estrogen replacement in a variety of health scenarios.
Dr Cauley: Between 2005 and 2007, we can expect a large amount of data to surface regarding breast cancer prevention, SERMs, and HRT. We will then have a lot more information on the safety of these interventions.
Dr Dore: If a woman does have some vaginal dryness and is on raloxifene, there are studies demonstrating that topical estrogen use is safe. Do you agree?
Dr Johnson: That's an important point. I believe that vaginal estrogen can be used in those women reasonably safely, now that we have available two forms of delivery systems that don't lead to systemic absorption and therefore don't lead to receptor competition. The estradiol ring (Estring) and estradiol vagina tablet (Vagifem) are not absorbed systemically and can be used safely in almost any patient if vaginal atrophy symptoms are present.
Dr Cauley: I just want to emphasize that raloxifene is not approved by the US Food and Drug Administration for breast cancer prevention. The data from the Multiple Outcomes of Raloxifene (MORE) study is, however, very exciting; it didn't demonstrate simulation of the endometrium with raloxifene. Other trials showed an increased risk of endometrial cancer with tamoxifen use, particularly in patients older than 50. Therefore, in women with a uterus, raloxifene has potential as an attractive therapy. Of course, we need more data before we can recommend it for breast cancer.
Dr Dore: Raloxifene is not recommended to control vasomotor symptoms, such as hot flashes, associated with menopause. However, it is a good option for asymptomatic women who want to remain on preventive therapy.
If a 70-year-old patient is using HRT and her BMD is high, would you recommend that she stop estrogen at that point because she's at increased risk for breast cancer?
Dr Cauley: The American College of Obstetrics and Gynecology recently recommended that after 5 years of HRT use, it is important to weigh the benefits and risks of HRT for an individual woman. Use of the Gail Model for Estimating Breast Cancer Risk can be helpful (Table). The major benefits of HRT are symptom relief and prevention of bone loss; and risks include possibly breast cancer, especially after long-term use and deep vein thrombosis. In other words, I think she needs to reconsider why she is taking the HRT.

Table 1. (click to enlarge) The Gail Model
Dr Johnson: For what reason is this 70-year-old patient continuing HRT? She clearly doesn't need it for bones. Perhaps it will help with Alzheimer's disease. She needs to understand that she is at somewhat increased risk for breast cancer, merely by being on the hormones for 20 years, let alone her high BMD. We have to be careful about how we explain those risks and benefits to patients.
As I understand it, the Alzheimer's-estrogen connection began about 10 years ago with a few very small open trials. Patients with Alzheimer's disease were given estrogen in an uncontrolled fashion and, after a few weeks, were observed by clinicians to be thinking better. Later, epidemiologic studies showed that women who had taken estrogen seemed to have about a 50% lower rate of developing Alzheimer's disease. Mechanistic studies in the lab showed that estrogen stimulated dendrite development. In theory, these factors explained why estrogen might help to prevent Alzheimer's disease in women. Two years ago, however, the first large, 1-year, randomized trial of estrogen vs placebo in a group of women with early Alzheimer's disease was conducted and showed no difference between the two groups. This study raised caution about the extent to which we should believe those epidemiologic studies; after all, they have the same kinds of biases with this disease as they do with others.
Dr Nolan: I've begun to focus more on study design and process, and when higher cortical function is observed, to examine the tests that are used and their reproducibility.
Dr Dore: Raloxifene is also being studied in the prevention and progression of Alzheimer's disease. One group looked at five different tests of cognitive functioning and showed that, using two of them, a significant improvement was seen.
Dr Nolan: I'd like to extend my thanks to everyone for joining me today and sharing your thoughts about postmenopausal health. This lively conversation is sure to have value to our readers in their pursuit of optimal patient care.
Faculty and Disclosures
Authors
|