
Figure 1. Sonographic appearance of the LNG-IUS. Sagittal view of the uterus: the proximal and distal ends of the vertical arm of the device, in the internal cervical os and the fundal region, respectively (arrows).


Figure 2. Sonographic appearance of the LNG-IUS. An acoustic
shadowing between both ends, representing the location of the levonorgestrel-coated
device (2A, Sagittal view; 2B, Transverse view.)
- Detection of the proximal and distal ends of the vertical arm of the
device, in the internal cervical os and the fundal region, respectively.
- An acoustic shadowing between both ends, representing the location of
the LNG-coated device. This acoustic shadowing is also demonstrated in the
transverse section (sagittal view - Figure 2A, transverse view - Figure
2B). By comparison, the sonographic appearance of the copper IUD is
completely echogenic (Figures 3A and 3B). It should be mentioned that the
string of the Mirena is much more echogenic than that of the T-380A device
marketed in the United States.
The introduction of LNG-IUS has brought a significant change in the
discontinuation rate of IUDs as a result of side effects. On the order of 20%
of conventional IUD users choose to have the device removed because of
increased menstrual blood loss and abnormal uterine bleeding.[4]
The LNG-IUS, on the other hand, can cause a dramatic reduction in blood loss[1]
and can even serve as an alternative approach to hysterectomy for the
treatment of menorrhagia.[5,6] In addition, the LNG-IUS can
significantly reduce blood loss and decrease the number of days of bleeding
per cycle.[7] Menorrhagia in conventional IUD users can be caused by decreased vascular
resistance in the uterine artery, with concomitant increased blood flow to the
uterus. These changes can be detected with transvaginal color Doppler
ultrasonography as changes in the pulsatility index (PI)[8,9] [see footnote*].
However, only a few studies have described the hemodynamic changes in LNG-IUS
users. Pakarinen and colleagues[10] examined the impedance to
uterine blood flow in 10 fertile women before and after the insertion of
LNG-IUD and did not find any change in the uterine PI. Jarvela and coworkers[11]
demonstrated that the LNG-IUS induces an increase in the main uterine artery
PI in the midluteal phase, with a concomitant decrease in serum progesterone
concentration. In addition, the extent of increase in the PI correlated with
the serum levonorgestrel concentration. Zalel and colleagues[12] have evaluated the uterine blood flow
in conventional IUD and LNG-IUS users and compared it with the clinical
patterns of the women carrying the device. Clinical measures of menstrual
bleeding, endometrial thickness, and Doppler flow of the cervical branch of
the uterine artery and spiral artery were examined. Forty-seven women carrying
LNG-IUS (group A) were compared with 52 women carrying copper IUD (group B,
defined as the control group). There was no significant difference between the
2 groups with regard to Doppler flow in the cervical branch of the uterine
artery (resistance index (RI) = 0.6 ± 0.01 in both groups, P = .9)
[see footnote†]. Endometrial width was
significantly thinner in group A (4.1 ± 0.2 mm) in comparison to group B (7.4
± 0.2 mm) (P < .0001). Subendometrial flow in the spiral artery was
significantly reduced in 35 women in group A (75%) and in none of the women in
group B (P < .0001) (Figures 4 and 5). Thus, the LNG-IUS did not
produce a significant change in the uterine artery blood flow (cervical
branch) relative to the copper IUD but did dramatically reduce the
subendometrial blood flow. Furthermore, this observation was reinforced by the
significant reduction in endometrial thickness in LNG-IUS users (from mean of
7.34 mm to 3.92 mm, P < .0001). This study offers an explanation for
the oligo-amenorrhea experienced by LNG-IUS users, which is that the device
induces a local progestational effect on the endometrium with no change in the
blood flow in the uterine artery. This conclusion is also supported by
findings of Xiao and colleagues,[13] who studied long-term use of
the LNG-IUS. Of note is that French and coworkers[14] have stated
that the amenorrhea in LNG-IUS users is an end-organ suppression of bleeding,
is benign, and is associated with normal estrogen levels.
After the demonstration of the local progestational effect of the LNG-IUS
on the endometrium, Zalel and colleagues evaluated the time required for this
effect to manifest (The progestational effect of the LNG-IUS - when does it is
manifest? Contraception, 2003, accepted for publication). Doppler flow
of the cervical branch of the uterine artery and spiral artery, as well as the
endometrial width (up to day 10 of the cycle), were evaluated in 36 women 1-2
months and 4-6 months after insertion of the LNG-IUS. During the first 2 months after insertion, 44% of women experienced
intermenstrual bleeding. After 4-6 months, however, only 8% of women
experienced intermenstrual bleeding. Complete cessation of menstrual bleeding
occurred in 5% of women 2 months after insertion and in 66% after 4-6 months.
Although there was no change in the Doppler flow in the cervical branch of the
uterine artery in either group, subendometrial flow in the spiral artery was
significantly reduced. This observation was reinforced by the demonstration of
significant reduction in the endometrial thickness after 4 months of use. This study has demonstrated that the concomitant reduction of clinically
intermenstrual bleeding as well as subendometrial blood flow and endometrial
thickness (ie, the local progestational effect of the levonorgestrel-releasing
IUS on the endometrium) are already manifest by the fourth month of use of the
LNG-IUS. The findings of this study are in accordance with those of Fraser and
coworkers [15] demonstrating that the breakthrough bleeding
experienced by many of the LNG-IUS users is especially relevant in the first
months after insertion of an IUD. The use of the LNG-IUS is frequently associated with follicular
dysfunction, which is related to circulating levonorgestrel concentrations.[16]
This dysfunction ranges from complete inhibition of ovulation[17]
to ovarian cyst formation.[18] The prevalence of the ovarian cysts
after exposure to levonorgestrel is well described, ranging from 12% to as
high as 73%.[19] However, little is known about the incidence of
these cysts after the insertion of an LNG-IUS, as detected by pelvic
ultrasonography. Zalel and Lidor (unpublished data, sent for publication) have performed a
prospective case-control study involving 106 women who were assessed for the
presence of ovarian cysts (> 25 mm) after the first menstrual period
following insertion of either an LNG-IUS (n = 53, the study group) or a copper
IUD (n = 53, the control group). The researchers found a significantly higher
incidence of ovarian cysts in the LNG-IUS group compared with the control
group (20.7% vs 7.5%, respectively, P < .0001). However, these
ovarian cysts seem to represent functional cysts, as they seldom exceeded 30
mm in diameter and assumed a benign ultrasonographic appearance; the RI
measurements of the cysts and the CA-125 levels were within the normal range.
Moreover, 40% of the cysts regressed within 6 months of follow-up and the rest
by the end of 1 year, a similar finding to that of Jarvela and colleagues,[20]
who reported that in their study, all of the ovarian cysts detected 3 months
after LNG-IUS insertion disappeared spontaneously within 4 months of
follow-up. A speculation to explain these cysts is that LNG exerts a direct
effect on the adjacent follicles via the blood circulation perfusing the
ovary.
The presented studies have shown the sonographic appearance of the LNG-IUS
in comparison to the copper IUD. It was also demonstrated that LNG-IUS has a
local progestational effect on the endometrium with no change in the blood
flow in the systemic circulation. Moreover, this effect is manifested in most
cases already on the fourth month after insertion of the device, the time
required for improvement of the intermenstrual bleeding. Furthermore, although
the use of LNG-IUS is associated with a high incidence of ovarian cysts
compared with the copper IUD, these cysts have benign sonographic, Doppler
flow, and laboratory characteristics and regress spontaneously during the
first months of follow-up.
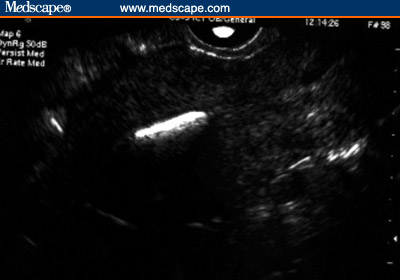
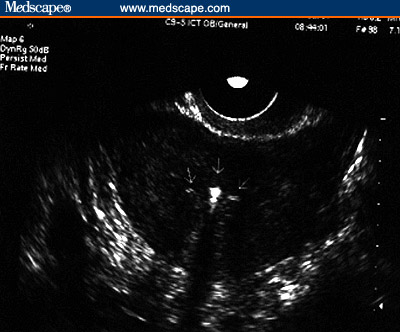
Figure 3. Copper IUD. (3A, Sagittal view [Note the
echogenicity of the device throughout its length)]; 3B, Transverse
view.)
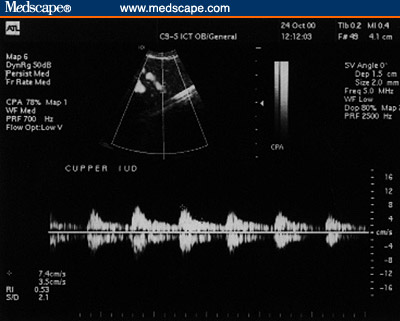
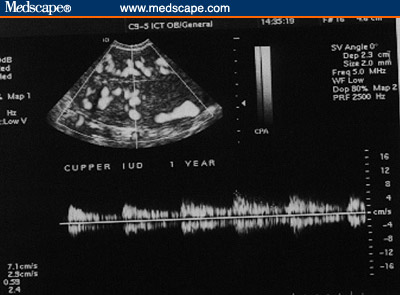
Figure 4. Copper IUD. Subendometrial flow. (4A, 1 month post
insertion; 4B, 1 year post-insertion [Note the rich flow even a year
post insertion of the IUD.])
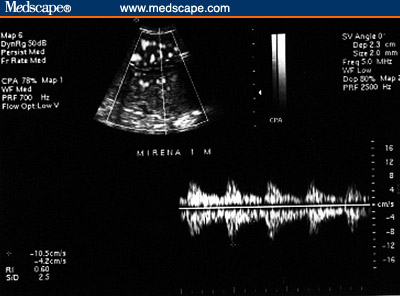
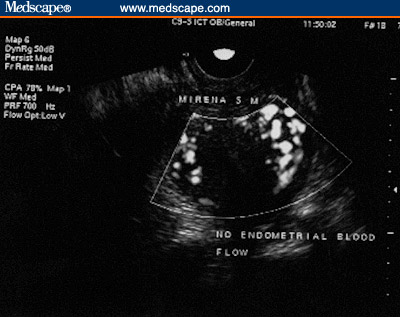
Figure 5. LNG-IUS. Subendometrial flow. (5A, 1 month
post-insertion [Note the rich flow]; 5B, 5 months post-insertion
[Note the lack of subendometrial flow]).
*Pulsatility index is defined as the following
ratio: systolic velocity – diastolic velocity/mean velocity.
†Resistance index is defined as the
ratio: systolic velocity – diastolic velocity/systolic velocity.
Summary
References
![]()
![]()