Ever wondered if things were not quite what they seem? That under a normal, fairly boring exterior, there is a second, weird level, full of mystery and unpredictability? Well, here you find something of that sort, and no, I'm not talking about Superman. I'm talking about things that are much smaller, so small in fact, we can hardly see them. And what are they? They are miniscule packets of energy, or quanta. The study of these particles is called quantum physics (k'wan-tum fees-iks). Quantum physics is weird. Actually, that is probably an understatement.
In fact, a very famous chemist and physicist, Neils Bohr, once remarked that “Anyone who isn't shocked by quantum theory has not understood it.”3 Originally introduced by Max Planck, quantum theory has been called the “most revolutionary idea which has ever shaken physics.2
Looking back on history and the development of quantum science, we can see quite clearly that it is perhaps one of the most revolutionary and paradigm-changing developments that scientists have ever encountered.9 
Neils Bohr

Max Planck
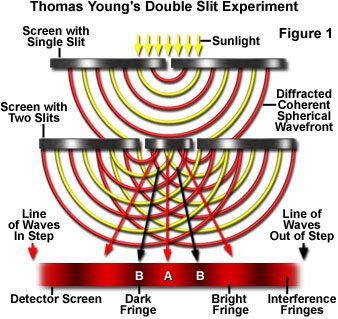
Thomas Young’s double slit experiment
Until the 1900s, Thomas Young's double slit experiment was regarded as conclusive proof that wave behaved as a particle. That was, until the problem of the photoelectric effect was brought into focus.
It had already been determined through experiments that “when light shines on a metal surface, the surface emits electrons”1 This of course, wasn't in itself proof that light travelled as a particle, since it was possible that if a wave of light hit an electron in one of the atoms of metal, it transferred energy to knock the electron out of its atom. This meant that the energy supposed to be contained in the waves excited electrons, causing them to “jump” out of the metal surface. 8 This would have made sense, if by increasing the intensity of the light, the electrons got more excited. The frequency of the light waves should not have affected the number of electrons emitted, and red and ultraviolet light of the same intensity should have knocked out the same number of electrons. Decreasing intensity should have meant decreasing the number of electrons knocked out of the metal also decreased. 8
Photoelectric Effect
It was at this point that a German experimenter, Philipp Lenard came in. Lenard studied the absorption of high-frequency light by certain metals, and in his experiments had used beams of monochromatic light (one color, single wavelength light), beaming them on metal strip and measuring the energy of the electrons ripped out of the metal. According to the above theory, his putting the light closer to the metal target should have meant the intensity had increased, resulting in more energy from the electrons that were being emitted, and their moving faster. Lenard found that this was not the case. An increase in the intensity meant that more electrons were released, but not that they moved any faster. This could only make sense if the light was travelling in particles. 2
This case was then supported by a formula that was developed by Max Plank. While Planck was working on the problem of electron radiation from metals, he did something no scientist had done before. He did not join up the energy portions. And yet his formula seemed to work perfectly with observations. 2 The rule that Planck came up with was one that stated the relationship between a small portion of energy, called a quantum, and the frequency of a wave. To determine the energy of one quantum, E, the frequency, f was multiplied by a constant, Planck's constant, with a value of energy of a quantum, E, one multiplies the wave frequency f by a fixed number, a constant, Planck's constant, that had a value of h=6.626 x 10-34 J•S 2 Although this was a fairly small constant, the idea suggested was an idea so radical not even Planck himself realized the significance of his discovery. Einstein, however, realized the significance of Planck's proposal, and proposed that light also delivers its energy in small packets that consist of very small quanta, called photons, each with an energy of Planck's constant times its frequency. In this case, it was shown that the frequency of light would indeed make a difference in the photoelectric effect. Higher frequency photons would have more energy, making electrons fly out of the metal faster. Experimentation seemed to agree with this proposal, and the photoelectric effect seemed to confirm that light travelled in particles. And therefore, it was fairly conclusively proven that light consisted of particles. 8 According to Einstein's final conclusion, each electron is knocked when hit by a photon of light, whose energy depends of the frequency of the light. Einstein argued that the reason we often don't observe the particle nature of light, but rather the wave nature is because of the extremely large number of photons in a small area, sort of the way we don't see individual dots of pixels on a printed image. 1 If you look very closely, or with a microscope at a printed paper, you will notice that all the print is made up of several very tiny dots. Now came the conclusion that light did indeed act as a particle. This, however, would make it extremely difficult to explain Young's double slit experiment. So, it was finally proposed by physicists that the nature of light was both that of a wave and particle, and this finally came to be known as the wave-particle duality of light, a nature soon observed in electrons, as well. 8 Studying electrons, later in history, it was revealed that it was not light alone that displayed this highly unpredictable behavior, but subatomic particles as well. 6
Werner Heisenberg
The second major principle in quantum physics is one suggested by Werner Heisenberg, called the principle of uncertainty. Heisenberg was the first person to realize the world runs on Quantum chance. 10 In 1925, when he was only 23 years old, Heisenberg published his theory of quantum mechanics. This theory won Heisenberg the Nobel Prize for Physics in 1932. 5 He is also famous for having proposed the uncertainty principle in 1927, at the age of 26. “It states that because matter is spread out in space, wavy, it is impossible to say exactly where it is and what it might do next simultaneously” 10 This basically meant that we could not know everything about the position AND velocity of an electron at the same time. The more precise the knowledge of the position of an electron was, the less precise the knowledge of the velocity of the electron, and vice versa. Therefore, if we know everything of the position of an electron, then we know nothing of the velocity and if we know everything about the velocity, then we know nothing of where the atom is. Also, it is almost impossible to predict where an electron will be in the next moment, if we know it's current location.
Heisenberg was one of the first to suggest that we cannot assign an electron a particular position in space at a certain time, nor can we follow its orbit. Heisenberg therefore concluded that the mechanical quantities that describe an electron, such as position and velocity could not be represented by ordinary numbers, but rather by abstract “matrices” which describe these quantities in terms of probability, or where the electron is most likely to be found in an atom. 5 In conclusion, quantum mechanics is a very dynamic and weird science, one that even today continues to baffle scientists in its utter unpredictability and apparent randomness. There are few things that can be said conclusively, and even then the conclusions scientists come to are fairly inconclusive in themselves, most conclusions being that nothing can be said conclusively. Quantum science was not only in itself a strange discovery, it demonstrated to scientists that no matter how much they find out about the universe, there is always more to learn, more that can astonish and amaze. This, of course means that scientists themselves will always learn new things. This can comfort students everywhere, in the fact that even the smartest people in the world have more to learn. 1) Al-Khalili, Jim. Quantum. London: Weidenfeld and Nicolson, 2003.2) Cline, Barbara L. Men Who Made a New Physics. New York: Cambridge UP, 1939.
3)Feynman, Richard P., Robert Leighton, and Matthew Sands. "What is Quantum Physics?" What is Quantum Physics. 12 Apr. 2009
4)Jones, Andrew Z. "Quantum Physics Overview." Quantum Physics- Overview of Quantum Physics. 2009. About.com. 12 Apr. 2009
5)McMahon, David. Quantum Field Theory Demystified. New York: Mcgraw Hill, 2008.
6)Nave, R. "Wave-Particle Duality." Wave-Particle Duality. Hyperphysics.phy-astr.gsu.edu. 12 Apr. 2009
7)Park, David. Introduction to Quantum Theory. Montreal: McGraw Hill, 1992.
8)Patel, Mahan. "Planck's Constant and the Energy of a Photon." Colorado University. 12 Apr. 2009
9)Rosenblum, Bruce, and Fred Kuttner. Quantum Enigma. New York: Oxford UP, 2006.
10)"YouTube - Quantum Mechanics for Dummies - Electrons Are Weird." YouTube - Broadcast Yourself. 19 Apr. 2009