(part 2 of "Aerobic stabilisation of pharmaceutical wastewaters using large scale extended aeration activated sludge process" by Saugath Lahiri..JNTUniv, Hyd, INDIA jan 2002)
.
MATERIALS AND METHODS:
MATERIALS:
The following materials and characteristics according to the specifications are given below were used in the study.
GLASSWARE:
The glassware used in the present study is of PYREX quality, manufactured by Borosil glassware LTD, MUMBAI.
WATER:
Water in all experiments was laboratory distilled water pH (7.2-8.0), alkalinity (40-120mg/l), and chlorides (20-30mg/l) for preparation of synthetic effluent and raw effluent dilution.
CHEMICALS:
All the chemicals used in the study were of analytical(AR) grade.
METHODS:
EFFLUENT CHARACTERISATION:
The raw effluent used for the study was characterized according to the standard methods suggested by APHA. The diluted effluent was prepared by diluting raw effluent to the required concentration. The raw effluent characteristics are given in the table xxx. All the analysis was in accordance with the APHA standards.
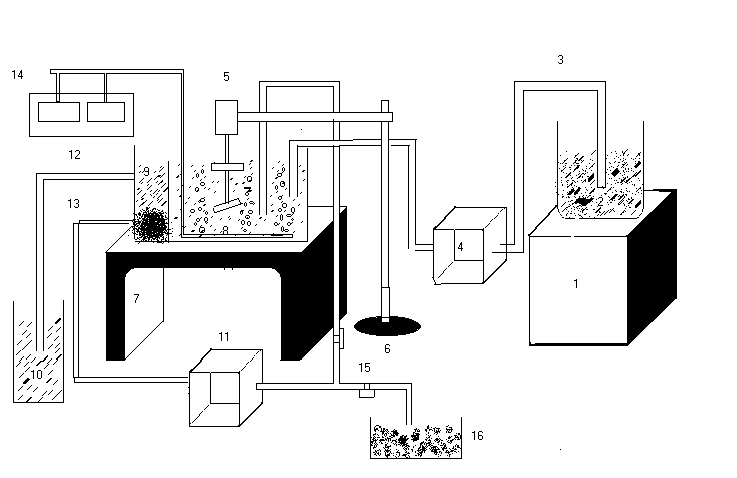
REACTOR CONFIGURATION:

HYDROGEN ION CONCENTRATION (pH):
The basic concentration of the electrometric pH determination of hydrogen ion concentration is potentiometric measurement using pH meter consisting of calomel glass electrode. Measurement of pH is one of the most important tests used in the chemical nature of effluent. The standard buffer solutions of pH values 4.0,7.0&9.2 are used for calibrating the instrument. Then the pH of the effluent was measured.
CONDUCTIVITY:
Conductivity is the capacity of water to carry an electrical charge, which depends on the total ionic concentration. Most of the total inorganic substances in water are in the ionized form and hence contribute to conductance. The conductivity of the effluent was determined at a temperature of 250C with the help of conductivity meter. The instrument was calibrated initially using 0.01N KCl to get the cell constant of 1413. Then the conductivity of the sample was measured.
SOLIDS:
Solids refer to matter suspended or dissolved in the effluent. Solid analysis is important in the control of biological and physical wastewater treatment processes. A well-mixed sample is evaporated in a weighed dish and dried to a constant weight in an oven at 103-1050. The increase in weight represents total solids. The difference gives the total suspended solids.
Total solids (mg/l) =[(A-B)/ml of sample]*1000 *1000
Where, A = weight of the dried residue +beaker in mg
And, B = weight of the empty beaker in mg
Suspended solids mg/l = TS (mg/l)-TDS (mg/l).
KJELDHAL NITROGEN:
In the presence of sulfuric acid and mercuric sulfate catalyst, originally bound nitrogen gas gets converted into ammonium sulfate. The digestate was made alkaline with NaoH and distilled. The liberated ammonia is absorbed in sulfuric acid of known concentration. The absorbed ammonia is determined by titrating the excess H2SO4 with standard alkali using methyl orange as an indicator.
Kjeldhal nitrogen can be calculated by using the formula
Nmg/l = (A-B)*1000*14*N/V
Where N= normality of H2SO4
A= volume of NaoH,
B= volume of NaoH consumed by the sample,
V = volume of the sample in ml.
CHLORIDES:
Chlorides are estimated by argentometric method. Chloride content of the sample is determined by titration with standard silver nitrate using potassium chromate as indicator. Chloride reacts with silver nitrate to form silver chloride. The excess silver reacts with chromate, which is brown, precipitate indicating the end point.
NITRATES:
Nitrogen sample is very important in connection with wastewater treatment. By controlling nitrification, aerobic treatment costs can be kept at a minimum. In the wastewater treatment systems, high amounts of nitrate denote the aerobic conditions and high solubility of the wastes.
Nitrates can be determined by phenol disulphonic acid to form a derivative, which in alkaline conditions develop yellow color. The intensity of the color is directly proportional to the concentration of nitrates in the sample.
Standards in the range were prepared from the standard nitrate solutions by evaporating it. 50ml of prepared standards were taken and evaporated to dryness. The residue left was dissolved with phenol disulphonic acid in the presence of sulfamic acid to obtain yellow color solution. 5ml of ammonium solution was added and then diluted to 100ml with distilled water. The absorbencies of the standards were recorded spectrophotometrically at 480nm. A standard graph of optical density Vs concentration was drawn. The sample was treated in a similar manner as standards and the optical density was recorded. The concentration of nitrate in the sample was obtained from the standard graphs.
PHOSPHATES:
Presence of phosphorous in the wastewater serves as an important nutrient for microorganisms useful in biological treatment of the waste. Many industrial wastes however do not contain sufficient quantities of phosphorous for optimum growth. In such cases additional inorganic phosphates may supply deficiencies.
The method adopted is spectrophotometric method, where in acidic conditions orthophosphates react with ammonium molybdate to form molybdophosphoric acid. SnCl2 reduces it to molybdenum blue. The intensity of the blue color complex is measured which is directly proportional to phosphate present in the sample.
Standard graph was drawn between concentration and absorbance at 690nm. The concentration of the phosphates was calculated from the standard graph.
SULPHATES:
Most of the salts of sulfates are soluble in water. Sulfates cause a problem of scaling in industries supplies and problem of odor and corrosion in wastewater treatment due to its reduction to H2S. Sulfates can be determined by turbidimetric method.
PRINCIPLE:
Sulfate ions are precipitated as BaSO4 in acidic media (HCl) with BaCl2. The absorption of this precipitated suspension is measured by spectrophotometer.
Ba2+ + SO42- BaSO4 (white precipitate)
An aliquot of diluted sample is taken and to 2.5 ml of the conditioning reagent was added and mixed thoroughly with the help of a magnetic stirrer. BaCl2 crystals are added while stirring. The reading is taken on a spectrophotometer at 420nm after 4min.
Standard graph is prepared between concentration and absorbance by carrying standard sulfate solution through procedure. The concentration of sulfate is known from the standard curve.
CHEMICAL OXYGEN DEMAND:
COD determines the O2 required for chemical oxidation of organic with the help of strong chemical oxidant under acidic conditions. The determination was done using HACH COD apparatus.
PRINCIPLE:
The test involves refluxing the sample with known amount k2Cr2O7 in the presence of concentrated H2SO4 followed by titration of the remaining dichromate, when oxidation was complete with a solution Fe2+ ions. The amount of dichromate consumed is related to the oxygen demand.
Cr2O7 + 14H+ 2Cr3+ + 7H2O
When chloride ions concentrate is high in used for COD determination cause serious problem because chloride ions utilizes the dichromate for its oxidation.
Cr2O7- + 6Cl- + 14H+ 3Cl2 + 3Cr3 + 7H2O
To remove chloride ions HgSO4 is added. The Hg2+ ion combines with chloride ions to form a poorly ionized HgCl2 complex, which is resistant to oxidation by dichromate. Straight chain aliphatic compounds and certain aromatic compounds are not fully oxidized that is why AgSO4 is added as a catalyst. In the present study, open reflux titrimetric method has been used.
PROCEDURE:
For COD analysis 20ml of the sample was taken in 250ml beaker. A pinch of HgSO4 was added into the beaker and to this 10ml of K2Cr2O7 and 30ml of sulfuric acid reagent was added. The beakers were covered with watch glass and kept on a hot plate and refluxed for 2hrs at 1500C for digestion. After refluxing the samples were cooled and the sides were washed to wash in the vapors. Then 2-3 drops of Ferroin indicator was added and titrated against standard 0.1N FAS solutions.
COD is calculated by using following formula:
CODmg/l = (A-B) * N *8 *1000 /V
Where A = FAS consumed
B = FAS consumed for sample
N = normality of FAS
V = volume of the sample taken in ml.
DISSOLVED OXYGEN:
This test gives the amount of oxygen present in dissolved state in a sample. The dissolved oxygen levels depend upon physical, chemical and biochemical activities. In aerobic biological treatment process the limited solubility of oxygen is of great importance. Because it governs the rate at which oxygen will be absorbed by the medium and therefore the cost of aeration. In the determination of DO, the MnSO4 in the alkaline conditions forms magnesium hydroxide, which is white precipitate. This reacts with oxygen to form brown hydrated oxide, in which Mn (II) is oxidized to Mn (IV). When sulfuric acid is added I- is oxidized to I2. The amount of I2 produced is equal to the initial amount of oxygen. This I2 is titrated with thiosulphate using starch as an indicator.
DO can be calculated by formula
1 ml of thiosulphate (0.0125N) =0.2 mg of O2
If Vsample is the volume of sample
and V the vol of thiosulphate consumed,
then the amount of oxygen present can be calculated as = V x 0.2
Vsample contains V X0.2 mg of O2
Hence Dissolved Oxygen DO mg/l = 0.2 * 1000 * V /Vsample
generally Vsample is taken as 200 ml.
BIOCHEMICAL OXYGEN DEMAND:
BOD denotes the amount of oxygen needed by microorganisms for stabilization of decomposable matter under aerobic conditions.
PROCEDURE: The samples are diluted with oxygenated water which contains trace amounts of nutrient material required by microbes (N,P,S,Fe,Ca) but which does not consume oxygen. The samples that are sterile require pre- treatment by filtering (because outlet samples contain mass) and seeding with bacteria. In all cases a blank is incubated consisting of only of only of the dilution water which has been subjected to the same pre-treatment as the samples.
Then for these samples initial DO is determined by winkler-Azide method and the samples are kept in BOD incubator for five days at 270C. During the incubation the DO decreases and the final DO5 was determined. The 5 days BOD at 270C is equal to the difference in the values.
BOD5 27 mg/l = (DO0 – DO5)
The BOD value is calculated by the following formula
BOD527 mg/l =Volume of the bottle XY / Volume of the sample in ml
Where,
Y =(DO0S – DO5S) –(DO0B – DO5B)
DO0S = initial DO of the sample
DO5S = final DO of the sample
DO0B = initial DO of the blank
DO5B = Final DO of the blank
MIXED LIQUOR SUSPENDED SOLIDS (MLSS):
The liquid suspension of microorganisms in aeration basin is generally referred to as mixed liquor and the biological growth are called mixed liquor suspended solids. In suspended growth systems, such as activated sludge, the organism concentration is usually referred to the mixed liquor suspended solids (MLSS) or its volatile fraction (MLVSS). This parameter is used in calculation of F/M ratio, SVI, Recirculation ratio, etc.
PROCEDURE:
Two beakers of 50ml were taken and weighed then known volume of mixed liquor from the aeration tank was taken. To one of the beaker the sample is completely transferred and labeled as TOTAL SOLIDS. Same amount of sample is withdrawn from the aeration tank and filtered through whatman filter paper No 42. The contents are evaporated on a hot plate and then in a hot air oven for about half-an-hour. The beakers are cooled in a dessicator and then the final weight was taken. The difference gives the MLSS.
MLSS mg/l = (A-B) * 1000 * 1000 / ml of the sample taken
where, A = weight of the total solids
B = weight of the total dissolved solids.
MIXED LIQUOR VOLATILE SUSPENDED SOLIDS:
There is evidence that active organisms compromise relatively small percentage of the total biological solids in conventional suspended growth systems. Measurements of gross organic content in the active solids, such as the MLVSS determination, therefore give an exaggerated measure of active microorganism’s content in the system. Nevertheless it has been found that there is usually a good correlation between total mass of MLVSS and other process parameters such as effluent BOD, rate of increase of biological solids and oxygen consumption. The ratio of the MLVSS to the MLSS tends to be relatively constant for a given system i.e. typically equivalent to about75-80%.
PROCEDURE: Whatman filter paper NO 42. Was taken and then known volume of mixed liquor from the aeration tank was taken and filtered. After filtration the initial weight of the crucible along with the filter paper was taken, then the crucible was kept in muffle furnace at 5500C for 15 minutes. After the crucible was removed from the furnace it was cooled in the dessicator, then the final weight of the crucible was taken. The difference including the MLVSS concentration was calculated using the following formula.
MLVSS mg/l = (A-B) * 1000 * 1000/ V
Where,
A = final weight of crucible in grams
B = initial weight of the crucible in grams
V = volume of the sample taken in ml.
OXYGEN UPTAKE RATE: (OUR)
Microbes in the activated sludge process use O2 as they utilize substrate in the waste. The rate at which they use O2, OUR, can be taken as a measure of the biological activity, low OUR’s indicate low biological activity. The use of OUR’s is also receiving recognition as a means of monitoring and controlling the ASP ( OUR is considered to be one of the important parameters in a aerobic system). OUR is mainly governed by oxygen concentration, oxidizable (reduced) substrate and physical and chemical properties of the waste and also the nature of the waste.
PROCEDURE:
The measurement of OUR was accomplished by using a DO probe with stirrer. One liter of the mixed liquor was taken in a beaker and aerated to reach the saturation DO level. Approximate fall in DO levels were recorded at fixed time intervals over a period of 45 minutes. The slope of the line from the plot of observed DO readings Vs time was taken as OUR.
SLUDGE VOLUME INDEX:
This is defined as the volume in ml occupied by one gram of activated sludge mixed liquor solids, dry weight, after settling for 30 minutes in a 1000ml graduated cylinder. SVI data yields information to establish the proper recirculation ratio for optimum process efficiency and maximum solids concentration in the waste sludge. SVI VALUES indicate the settling characteristics of the sludge.
50 --------- Excellent sludge
50-100 --------- Good
100-150 --------- satisfactory
150 or above indicates the bulking of sludge.
PROCEDURE:
SVI was measured by taking one liter of the mixed liquor from the aeration tank and it is allowed to settle for 30 minutes in 100ml cylinder. The volume occupied by the sludge is measured. The SVI is calculated by
SVI = (Settled volume of the sludge after 30 min (ml/lit)) *1000 ml / MLSS concentration in mg/l of the sludge sample before settling
SLUDGE CONDITION:
It is useful in the estimation of process of unit efficiency. It can be studied by
J Appearance [Brown]
J Settleability [SVI]
FORWARD to part 3 Manufacturing processes in pharma industry